Essential role of voltage-dependent anion channel in various forms of apoptosis in mammalian cells
- PMID: 11266442
- PMCID: PMC2199613
- DOI: 10.1083/jcb.152.2.237
Essential role of voltage-dependent anion channel in various forms of apoptosis in mammalian cells
Abstract
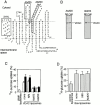
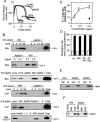
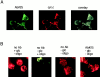
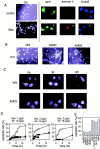
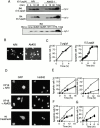
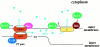
Through direct interaction with the voltage-dependent anion channel (VDAC), proapoptotic members of the Bcl-2 family such as Bax and Bak induce apoptogenic cytochrome c release in isolated mitochondria, whereas BH3-only proteins such as Bid and Bik do not directly target the VDAC to induce cytochrome c release. To investigate the biological significance of the VDAC for apoptosis in mammalian cells, we produced two kinds of anti-VDAC antibodies that inhibited VDAC activity. In isolated mitochondria, these antibodies prevented Bax-induced cytochrome c release and loss of the mitochondrial membrane potential (Deltapsi), but not Bid-induced cytochrome c release. When microinjected into cells, these anti-VDAC antibodies, but not control antibodies, also prevented Bax-induced cytochrome c release and apoptosis, whereas the antibodies did not prevent Bid-induced apoptosis, indicating that the VDAC is essential for Bax-induced, but not Bid-induced, apoptogenic mitochondrial changes and apoptotic cell death. In addition, microinjection of these anti-VDAC antibodies significantly inhibited etoposide-, paclitaxel-, and staurosporine-induced apoptosis. Furthermore, we used these antibodies to show that Bax- and Bak-induced lysis of red blood cells was also mediated by the VDAC on plasma membrane. Taken together, our data provide evidence that the VDAC plays an essential role in apoptogenic cytochrome c release and apoptosis in mammalian cells.
Figures


References
-
- Adams J.M., Cory S. The Bcl-2 protein familyarbiters of cell survival. Science. 1998;281:1322–1326. - PubMed
-
- Antonsson B., Conti F., Ciavatta A., Montessuit S., Lewis S., Martinou I., Bernasconi L., Bernard A., Mermod J.J., Mazzei G. Inhibition of Bax channel-forming activity by Bcl-2. Science. 1997;277:370–372. - PubMed
-
- Babel D., Walter G., Gøtz H., Thinnes F.P., Jürgens L., Kønig U., Hilschmann N. Studies on human porin. VI. Production and characterization of eight monoclonal mouse antibodies against the human VDAC “Porin 31HL” and their application for histotopological studies in human skeletal muscle. Biol. Chem. Hoppe Seyler. 1991;372:1027–1034. - PubMed
-
- Benz R., Maier E., Thinnes F.P., Gøtz H., Hilschmann N. Studies on human porin. VII. The channel properties of the human B-lymphocyte membrane-derived “Porin 31HL” are similar to those of mitochondrial porins. Biol. Chem. Hoppe Seyler. 1992;373:295–303. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

