Chunnel vision. Export and efflux through bacterial channel-tunnels
- PMID: 11269495
- PMCID: PMC1083749
- DOI: 10.1093/embo-reports/kvd075
Chunnel vision. Export and efflux through bacterial channel-tunnels
Abstract
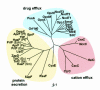
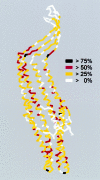
The Escherichia coli TolC protein is central to toxin export and drug efflux across the inner and outer cell membranes and the intervening periplasmic space. The crystal structure has revealed that TolC assembles into a remarkable alpha-helical trans-periplasmic cylinder (tunnel) embedded in the outer membrane by a contiguous beta-barrel (channel), so providing a large duct open to the outside environment. The channel-tunnel structure is conserved in TolC homologues throughout Gram-negative bacteria, and it is envisaged that they are recruited and opened, through a common mechanism, by substrate-specific inner-membrane complexes.
Figures


References
-
- Benz R., Maier, E. and Gentschev, I. (1993) TolC of Escherichia coli functions as an outer membrane channel. Zentralbl. Bakteriol., 278, 187–196. - PubMed
-
- Buchanan S.K, Smith, B.S., Venkatramani, L., Xia, D., Esser, L., Palnitkar, M., Chakraborty, R., van der Helm, D. and Deisenhofer, J. (1999) Crystal structure of the outer membrane active transporter FepA from Escherichia coli. Nature Struct. Biol., 6, 56–63. - PubMed
-
- Cowan S.W., Schirmer, T., Rummel, G., Steiert, M., Ghosh, R., Pauptit, R.A., Jansonius, J.N. and Rosenbusch, J.P. (1992) Crystal structures explain functional properties of two E.coli porins. Nature, 358, 727–733. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

