Glucocorticoids attenuate T cell receptor signaling
- PMID: 11283153
- PMCID: PMC2193373
- DOI: 10.1084/jem.193.7.803
Glucocorticoids attenuate T cell receptor signaling
Abstract
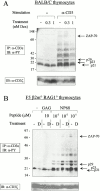
Glucocorticoids (GCs) affect peripheral immune responses by inhibiting T cell immunity at several stages of the activation cascade, causing impaired cytokine production and effector function. The recent demonstration that the thymic epithelium and possibly thymocytes themselves produce steroids suggests that endogenous GCs also play a role in the control of T cell development. As both peripheral responsiveness and thymic differentiation appear to be regulated by the quantity and quality of intracellular signals issued by antigen-major histocompatibility complex-engaged T cell receptor (TCR) complexes, we investigated the effects of GCs on the signaling properties of T cells stimulated by anti-CD3 monoclonal antibodies or agonist peptides. We demonstrate in this work that dexamethasone, a synthetic GC, inhibits the early signaling events initiated upon TCR ligation, such as tyrosine phosphorylation of several TCR-associated substrates including the zeta chain, the ZAP70 kinase, and the transmembrane adapter molecule linker for activation of T cells. Hypophosphorylation was not a consequence of reduced kinase activity of src protein tyrosine kinases, but was correlated with an altered- membrane compartmentalization of these molecules. These observations indicate that in addition to their well-described ability to interfere with the transcription of molecules involved in peripheral responses, GCs inhibit T cell activation by affecting the early phosphorylating events induced after TCR ligation.
Figures




Similar articles
-
The immunosuppressive effect of glucocorticoids in human primary T cells is mainly mediated via a rapid inhibition of the IL-2/IL-2R signaling axis.Cell Commun Signal. 2025 Jun 4;23(1):268. doi: 10.1186/s12964-025-02266-0. Cell Commun Signal. 2025. PMID: 40468327 Free PMC article.
-
Transcriptional regulation of kinases downstream of the T cell receptor: another immunomodulatory mechanism of glucocorticoids.BMC Pharmacol Toxicol. 2014 Jul 3;15:35. doi: 10.1186/2050-6511-15-35. BMC Pharmacol Toxicol. 2014. PMID: 24993777 Free PMC article.
-
Rescue of thymocytes and T cell hybridomas from glucocorticoid-induced apoptosis by stimulation via the T cell receptor/CD3 complex: a possible in vitro model for positive selection of the T cell repertoire.Eur J Immunol. 1991 Mar;21(3):643-8. doi: 10.1002/eji.1830210316. Eur J Immunol. 1991. PMID: 1826261
-
Glucocorticoid signaling: a nongenomic mechanism for T-cell immunosuppression.Trends Mol Med. 2007 Apr;13(4):158-63. doi: 10.1016/j.molmed.2007.02.001. Epub 2007 Feb 9. Trends Mol Med. 2007. PMID: 17293163 Review.
-
Glucocorticoids in T cell development, differentiation and function.Nat Rev Immunol. 2021 Apr;21(4):233-243. doi: 10.1038/s41577-020-00464-0. Epub 2020 Nov 4. Nat Rev Immunol. 2021. PMID: 33149283 Review.
Cited by
-
Discovery of novel irreversible inhibitors of interleukin (IL)-2-inducible tyrosine kinase (Itk) by targeting cysteine 442 in the ATP pocket.J Biol Chem. 2013 Sep 27;288(39):28195-206. doi: 10.1074/jbc.M113.474114. Epub 2013 Aug 9. J Biol Chem. 2013. PMID: 23935099 Free PMC article.
-
Glucocorticoid-mediated inhibition of Lck modulates the pattern of T cell receptor-induced calcium signals by down-regulating inositol 1,4,5-trisphosphate receptors.J Biol Chem. 2009 Nov 13;284(46):31860-71. doi: 10.1074/jbc.M109.005579. Epub 2009 Sep 23. J Biol Chem. 2009. PMID: 19776014 Free PMC article.
-
Adaptive immunity, chronic inflammation and the clock.Semin Immunopathol. 2022 Mar;44(2):209-224. doi: 10.1007/s00281-022-00919-7. Epub 2022 Mar 1. Semin Immunopathol. 2022. PMID: 35233691 Free PMC article. Review.
-
Impact of systemic steroids on the efficacy of first line imatinib treatment of patients with advanced gastrointestinal stromal tumors (GISTs).BMC Cancer. 2024 Oct 8;24(1):1245. doi: 10.1186/s12885-024-13032-9. BMC Cancer. 2024. PMID: 39379868 Free PMC article.
-
Redefining the human corneal immune compartment using dynamic intravital imaging.Proc Natl Acad Sci U S A. 2023 Aug;120(31):e2217795120. doi: 10.1073/pnas.2217795120. Epub 2023 Jul 24. Proc Natl Acad Sci U S A. 2023. PMID: 37487076 Free PMC article.
References
-
- Germain R.N., Stefanova I. The dynamics of T cell receptor signalingcomplex orchestration and the key roles of tempo and cooperation. Annu. Rev. Immunol. 1999;17:467–522. - PubMed
-
- Constant S.L., Bottomly K. Induction of Th1 and Th2 CD4+ T cell responsesthe alternative approaches. Annu. Rev. Immunol. 1997;15:297–322. - PubMed
-
- Kersh E.N., Shaw A.S., Allen P.M. Fidelity of T cell activation through multistep T cell receptor ζ phosphorylation. Science. 1998;281:572–575. - PubMed
-
- Evavold B.D., Sloan-Lancaster J., Allen P.M. Tickling the TCRselective T-cell functions stimulated by altered peptide ligands. Immunol. Today. 1993;14:602–609. - PubMed
-
- Sloan-Lancaster J., Allen P.M. Altered peptide ligand-induced partial T cell activationmolecular mechanisms and role in T cell biology. Annu. Rev. Immunol. 1996;14:1–27. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

