Functional reorganization of sensory pathways in the rat spinal dorsal horn following peripheral nerve injury
- PMID: 11283238
- PMCID: PMC2278535
- DOI: 10.1111/j.1469-7793.2001.0241g.x
Functional reorganization of sensory pathways in the rat spinal dorsal horn following peripheral nerve injury
Abstract
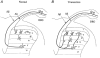
Functional reorganization of sensory pathways in the rat spinal dorsal horn following sciatic nerve transection was examined using spinal cord slices with an attached dorsal root. Slices were obtained from animals whose sciatic nerve had been transected 2-4 weeks previously and compared to sham-operated controls. Whole-cell recordings from substantia gelatinosa neurones in sham-operated rats, to which nociceptive information was preferentially transmitted, revealed that dorsal root stimulation sufficient to activate A afferent fibres evoked a mono- and/or polysynaptic EPSC in 111 of 131 (approximately 85%) neurones. This is in contrast to the response following A fibre stimulation, where monosynaptic EPSCs were observed in 2 of 131 (approximately 2%) neurones and polysynaptic EPSCs were observed in 18 of 131 (approximately 14%) neurones. In sciatic nerve-transected rats, however, a polysynaptic EPSC following stimulation of A afferents was elicited in 30 of 37 (81%) neurones and a monosynaptic EPSC evoked by A afferent stimulation was detected in a subset of neurones (4 of 37, approximately 11%). These observations suggest that, following sciatic nerve transection, large myelinated A afferent fibres establish synaptic contact with interneurones and transmit innocuous information to substantia gelatinosa. This functional reorganization of the sensory circuitry may constitute an underlying mechanism, at least in part, for sensory abnormalities following peripheral nerve injuries.
Figures
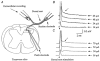
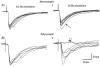

 ). Most neurones in SNT rats responded at a stimulus intensity less than the Aδ afferent threshold (< 33 μA).
). Most neurones in SNT rats responded at a stimulus intensity less than the Aδ afferent threshold (< 33 μA).
Similar articles
-
Peripheral nerve injury alters excitatory synaptic transmission in lamina II of the rat dorsal horn.J Physiol. 2003 Apr 1;548(Pt 1):131-8. doi: 10.1113/jphysiol.2002.036186. Epub 2003 Feb 7. J Physiol. 2003. PMID: 12576493 Free PMC article.
-
Synaptic responses of substantia gelatinosa neurones to dorsal column stimulation in rat spinal cord in vitro.J Physiol. 1994 Jul 1;478 ( Pt 1)(Pt 1):87-99. doi: 10.1113/jphysiol.1994.sp020232. J Physiol. 1994. PMID: 7965839 Free PMC article.
-
Alteration in synaptic inputs through C-afferent fibers to substantia gelatinosa neurons of the rat spinal dorsal horn during postnatal development.Neuroscience. 2000;99(3):549-56. doi: 10.1016/s0306-4522(00)00224-4. Neuroscience. 2000. PMID: 11029546
-
Pathobiological reactions of C-fibre primary sensory neurones to peripheral nerve injury.Exp Physiol. 1992 May;77(3):405-31. doi: 10.1113/expphysiol.1992.sp003603. Exp Physiol. 1992. PMID: 1321641 Review.
-
Spinal cord mechanisms of pain.Br J Anaesth. 2008 Jul;101(1):8-16. doi: 10.1093/bja/aen088. Epub 2008 Apr 15. Br J Anaesth. 2008. PMID: 18417503 Review.
Cited by
-
Actions of brain-derived neurotrophic factor on spinal nociceptive transmission during inflammation in the rat.J Physiol. 2005 Dec 1;569(Pt 2):685-95. doi: 10.1113/jphysiol.2005.095331. Epub 2005 Oct 6. J Physiol. 2005. PMID: 16210356 Free PMC article.
-
Role of presynaptic muscarinic and GABA(B) receptors in spinal glutamate release and cholinergic analgesia in rats.J Physiol. 2002 Sep 15;543(Pt 3):807-18. doi: 10.1113/jphysiol.2002.020644. J Physiol. 2002. PMID: 12231640 Free PMC article.
-
Stress Increases Peripheral Axon Growth and Regeneration through Glucocorticoid Receptor-Dependent Transcriptional Programs.eNeuro. 2017 Aug 21;4(4):ENEURO.0246-17.2017. doi: 10.1523/ENEURO.0246-17.2017. eCollection 2017 Jul-Aug. eNeuro. 2017. PMID: 28828403 Free PMC article.
-
Organization of intralaminar and translaminar neuronal connectivity in the superficial spinal dorsal horn.J Neurosci. 2009 Apr 22;29(16):5088-99. doi: 10.1523/JNEUROSCI.6175-08.2009. J Neurosci. 2009. PMID: 19386904 Free PMC article.
-
Expression of TRPV1 channels after nerve injury provides an essential delivery tool for neuropathic pain attenuation.PLoS One. 2012;7(9):e44023. doi: 10.1371/journal.pone.0044023. Epub 2012 Sep 4. PLoS One. 2012. PMID: 22962595 Free PMC article.
References
-
- Basbaum AI, Chi S-I, Levine JD. Peripheral and central contribution to the persistent expression of the c-fos proto-oncogene in spinal cord after peripheral nerve injury. In: Willis WD, editor. Hyperalgesia and Allodynia. New York: Raven Press, Ltd; 1992. pp. 295–304.
-
- Beal JA, Bicknell HR., Jr . Development and maturation of neurons in the substantia gelatinosa (SG) of the rat spinal cord. In: Rowe M, Willis WD Jr, editors. Development, Organization and Processing in Somatosensory Pathway. New York: Wiley-Liss; 1985. pp. 23–30.
-
- Bennett GJ, Laird JMA. Central changes contributing to neuropathic hyperalgesia. In: Willis WD, editor. Hyperalgesia and Allodynia. New York: Raven Press, Ltd; 1992. pp. 305–310.
-
- Blumenkopf B, Lipman JJ. Studies in autotomy: its pathophysiology and usefulness as a model of chronic pain. Pain. 1992;45:203–209. - PubMed
-
- Carlson J, Lais AC, Dyck PJ. Axonal atrophy from permanent peripheral axotomy in adult cat. Journal of Neuropathology and Experimental Neurology. 1979;38:579–585. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

