GPI anchoring leads to sphingolipid-dependent retention of endocytosed proteins in the recycling endosomal compartment
- PMID: 11285223
- PMCID: PMC145477
- DOI: 10.1093/emboj/20.7.1583
GPI anchoring leads to sphingolipid-dependent retention of endocytosed proteins in the recycling endosomal compartment
Abstract
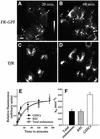
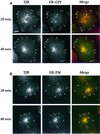
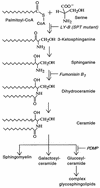
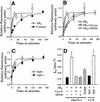
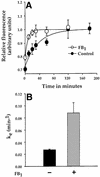
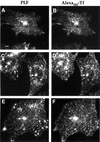
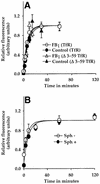
Glycosylphosphatidylinositol (GPI) anchoring is important for the function of several proteins in the context of their membrane trafficking pathways. We have shown previously that endocytosed GPI-anchored proteins (GPI-APs) are recycled to the plasma membrane three times more slowly than other membrane components. Recently, we found that GPI-APs are delivered to endocytic organelles, devoid of markers of the clathrin-mediated pathway, prior to their delivery to a common recycling endosomal compartment (REC). Here we show that the rate-limiting step in the recycling of GPI-APs is their slow exit from the REC; replacement of the GPI anchor with a transmembrane protein sequence abolishes retention in this compartment. Depletion of endogenous sphingolipid levels using sphingolipid synthesis inhibitors or in a sphingolipid-synthesis mutant cell line specifically enhances the rate of endocytic recycling of GPI-APs to that of other membrane components. We have shown previously that endocytic retention of GPI-APs is also relieved by cholesterol depletion. These findings strongly suggest that functional retention of GPI-APs in the REC occurs via their association with sphingolipid and cholesterol-enriched sorting platforms or 'rafts'.
Figures
References
-
- Ariga T., Macala,L.J., Saito,M., Margolis,R.K., Greene,L.A., Margolis,R.U. and Yu,R.K. (1988) Lipid composition of PC12 pheochromocytoma cells: characterization of globoside as a major neutral glycolipid. Biochemistry, 27, 52–58. - PubMed
-
- Bamezai A., Goldmacher,V.S. and Rock,K.L. (1992) Internalization of glycosyl-phosphatidylinositol (GPI)-anchored lymphocyte proteins. II. GPI-anchored and transmembrane molecules internalize through distinct pathways. Eur. J. Immunol., 22, 15–21. - PubMed
-
- Bielawska A., Crane,H.M., Liotta,D., Obeid,L.M. and Hannun,Y.A. (1993) Selectivity of ceramide-mediated biology. Lack of activity of erythro-dihydroceramide. J. Biol. Chem., 268, 26226–26232. - PubMed
-
- Brown D.A. and London,E. (1998a) Functions of lipid rafts in biological membranes. Annu. Rev. Cell. Dev. Biol., 14, 111–136. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

