Donor-derived IP-10 initiates development of acute allograft rejection
- PMID: 11304558
- PMCID: PMC2193411
- DOI: 10.1084/jem.193.8.975
Donor-derived IP-10 initiates development of acute allograft rejection
Abstract
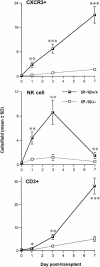
An allograft is often considered an immunologically inert playing field on which host leukocytes assemble and wreak havoc. However, we demonstrate that graft-specific physiologic responses to early injury initiate and promulgate destruction of vascularized grafts. Serial analysis of allografts showed that intragraft expression of the three chemokine ligands for the CXC chemo-kine receptor CXCR3 was induced in the order of interferon (IFN)-gamma-inducible protein of 10 kD (IP-10, or CXCL10), IFN-inducible T cell alpha-chemoattractant (I-TAC; CXCL11), and then monokine induced by IFN-gamma (Mig, CXCL9). Initial IP-10 production was localized to endothelial cells, and only IP-10 was induced by isografting. Anti-IP-10 monoclonal antibodies prolonged allograft survival, but surprisingly, IP-10-deficient (IP-10(-/-)) mice acutely rejected allografts. However, though allografts from IP-10(+/+) mice were rejected by day 7, hearts from IP-10(-/-) mice survived long term. Compared with IP-10(+/+) donors, use of IP-10(-/-) donors reduced intragraft expression of cytokines, chemokines and their receptors, and associated leukocyte infiltration and graft injury. Hence, tissue-specific generation of a single chemokine in response to initial ischemia/reperfusion can initiate progressive graft infiltration and amplification of multiple effector pathways, and targeting of this proximal chemokine can prevent acute rejection. These data emphasize the pivotal role of donor-derived IP-10 in initiating alloresponses, with implications for tissue engineering to decrease immunogenicity, and demonstrate that chemokine redundancy may not be operative in vivo.
Figures









References
-
- Wieder K.J., Hancock W.W., Schmidbauer G., Corpier C.L., Wieder I., Kobzik L., Strom T.B., Kupiec-Weglinski J.W. Rapamycin treatment depresses intragraft expression of KC/MIP-2, granzyme B, and IFN-γ in rat recipients of cardiac allografts. J. Immunol. 1993;151:1158–1166. - PubMed
-
- Hancock W.W., Gao W., Faia K.L., Csizmadia V. Chemokines and their receptors in allograft rejection. Curr. Opin. Immunol. 2000;12:511–516. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

