Discordant effects of anti-VLA-4 treatment before and after onset of relapsing experimental autoimmune encephalomyelitis
- PMID: 11306603
- PMCID: PMC199558
- DOI: 10.1172/JCI11717
Discordant effects of anti-VLA-4 treatment before and after onset of relapsing experimental autoimmune encephalomyelitis
Abstract
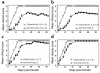
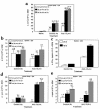
Initial migration of encephalitogenic T cells to the central nervous system (CNS) in relapsing experimental autoimmune encephalomyelitis (R-EAE), an animal model of multiple sclerosis (MS), depends on the interaction of the alpha4 integrin (VLA-4) expressed on activated T cells with VCAM-1 expressed on activated cerebrovascular endothelial cells. Alternate homing mechanisms may be employed by infiltrating inflammatory cells after disease onset. We thus compared the ability of anti-VLA-4 to regulate proteolipid protein (PLP) 139-151-induced R-EAE when administered either before or after disease onset. Preclinical administration of anti-VLA-4 either to naive recipients of primed encephalitogenic T cells or to mice 1 week after peptide priming, i.e., before clinical disease onset, inhibited the onset and severity of clinical disease. In contrast, Ab treatment either at the peak of acute disease or during remission exacerbated disease relapses and increased the accumulation of CD4(+) T cells in the CNS. Most significantly, anti-VLA-4 treatment either before or during ongoing R-EAE enhanced Th1 responses to both the priming peptide and endogenous myelin epitopes released secondary to acute tissue damage. Collectively, these results suggest that treatment with anti-VLA-4 Ab has multiple effects on the immune system and may be problematic in treating established autoimmune diseases such as MS.
Figures





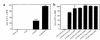
Similar articles
-
Differential effects of treatment with a small-molecule VLA-4 antagonist before and after onset of relapsing EAE.Blood. 2003 Dec 15;102(13):4464-71. doi: 10.1182/blood-2003-03-0974. Epub 2003 Aug 21. Blood. 2003. PMID: 12933585
-
Differential adhesion molecule requirements for immune surveillance and inflammatory recruitment.Brain. 2000 Jun;123 ( Pt 6):1092-101. doi: 10.1093/brain/123.6.1092. Brain. 2000. PMID: 10825349
-
The development of experimental autoimmune encephalomyelitis in the mouse requires alpha4-integrin but not alpha4beta7-integrin.J Clin Invest. 1998 Dec 15;102(12):2096-105. doi: 10.1172/JCI4271. J Clin Invest. 1998. PMID: 9854045 Free PMC article.
-
Evidence that Fas and FasL contribute to the pathogenesis of experimental autoimmune encephalomyelitis.Arch Immunol Ther Exp (Warsz). 2000;48(5):381-8. Arch Immunol Ther Exp (Warsz). 2000. PMID: 11140465 Review.
-
Anti-alpha4 integrin therapy for multiple sclerosis: mechanisms and rationale.Neurology. 2005 Apr 26;64(8):1336-42. doi: 10.1212/01.WNL.0000158329.30470.D0. Neurology. 2005. PMID: 15851719 Review.
Cited by
-
B-cell very late antigen-4 deficiency reduces leukocyte recruitment and susceptibility to central nervous system autoimmunity.Ann Neurol. 2015 May;77(5):902-8. doi: 10.1002/ana.24387. Epub 2015 Mar 28. Ann Neurol. 2015. PMID: 25712734 Free PMC article.
-
[Blocking adhesion molecules with natalizumab in multiple sclerosis].Nervenarzt. 2005 Aug;76(8):999-1005. doi: 10.1007/s00115-005-1900-2. Nervenarzt. 2005. PMID: 15812675 Review. German.
-
CNS elevation of vascular and not mucosal addressin cell adhesion molecules in patients with multiple sclerosis.Am J Pathol. 2010 Feb;176(2):556-62. doi: 10.2353/ajpath.2010.090437. Epub 2009 Dec 24. Am J Pathol. 2010. PMID: 20035048 Free PMC article.
-
Suppression of experimental autoimmune encephalomyelitis by extracellular adherence protein of Staphylococcus aureus.J Exp Med. 2006 Apr 17;203(4):985-94. doi: 10.1084/jem.20051681. Epub 2006 Apr 3. J Exp Med. 2006. PMID: 16585266 Free PMC article.
-
Experimental autoimmune encephalomyelitis (EAE) as a model for multiple sclerosis (MS).Br J Pharmacol. 2011 Oct;164(4):1079-106. doi: 10.1111/j.1476-5381.2011.01302.x. Br J Pharmacol. 2011. PMID: 21371012 Free PMC article. Review.
References
-
- Brown AM, McFarlin DE. Relapsing experimental allergic encephalomyelitis in the SJL/J mouse. Lab Invest. 1981;45:278–284. - PubMed
-
- McRae BL, Kennedy MK, Tan LJ, Dal Canto MC, Miller SD. Induction of active and adoptive chronic-relapsing experimental autoimmune encephalomyelitis (EAE) using an encephalitogenic epitope of proteolipid protein. J Neuroimmunol. 1992;38:229–240. - PubMed
-
- Lehmann PV, Sercarz EE, Forsthuber T, Dayan CM, Gammon G. Determinant spreading and the dynamics of the autoimmune T-cell repertoire. Immunol Today. 1993;14:203–208. - PubMed
-
- Vanderlugt CL, Karandikar NJ, Bluestone JA, Miller SD. The functional significance of epitope spreading and its regulation by costimulatory interactions. Immunol Rev. 1998;164:63–72. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

