Radial and longitudinal diffusion of myoglobin in single living heart and skeletal muscle cells
- PMID: 11320218
- PMCID: PMC33311
- DOI: 10.1073/pnas.101109798
Radial and longitudinal diffusion of myoglobin in single living heart and skeletal muscle cells
Abstract
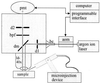
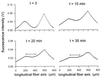
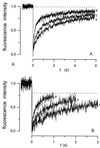
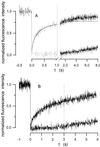
We have used a fluorescence recovery after photobleaching (FRAP) technique to measure radial diffusion of myoglobin and other proteins in single skeletal and cardiac muscle cells. We compare the radial diffusivities, D(r) (i.e., diffusion perpendicular to the long fiber axis), with longitudinal ones, D(l) (i.e., parallel to the long fiber axis), both measured by the same technique, for myoglobin (17 kDa), lactalbumin (14 kDa), and ovalbumin (45 kDa). At 22 degrees C, D(l) for myoglobin is 1.2 x 10(-7) cm(2)/s in soleus fibers and 1.1 x 10(-7) cm(2)/s in cardiomyocytes. D(l) for lactalbumin is similar in both cell types. D(r) for myoglobin is 1.2 x 10(-7) cm(2)/s in soleus fibers and 1.1 x 10(-7) cm(2)/s in cardiomyocytes and, again, similar for lactalbumin. D(l) and D(r) for ovalbumin are 0.5 x 10(-7) cm(2)/s. In the case of myoglobin, both D(l) and D(r) at 37 degrees C are about 80% higher than at 22 degrees C. We conclude that intracellular diffusivity of myoglobin and other proteins (i) is very low in striated muscle cells, approximately 1/10 of the value in dilute protein solution, (ii) is not markedly different in longitudinal and radial direction, and (iii) is identical in heart and skeletal muscle. A Krogh cylinder model calculation holding for steady-state tissue oxygenation predicts that, based on these myoglobin diffusivities, myoglobin-facilitated oxygen diffusion contributes 4% to the overall intracellular oxygen transport of maximally exercising skeletal muscle and less than 2% to that of heart under conditions of high work load.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

