Cisplatinum and taxol induce different patterns of p53 phosphorylation
- PMID: 11326311
- PMCID: PMC1505020
- DOI: 10.1038/sj.neo.7900122
Cisplatinum and taxol induce different patterns of p53 phosphorylation
Abstract
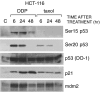
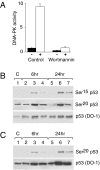
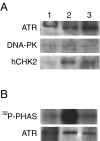
Posttranslational modifications of p53 induced by two widely used anticancer agents, cisplatinum (DDP) and taxol were investigated in two human cancer cell lines. Although both drugs were able to induce phosphorylation at serine 20 (Ser20), only DDP treatment induced p53 phosphorylation at serine 15 (Ser15). Moreover, both drug treatments were able to increase p53 levels and consequently the transcription of waf1 and mdm-2 genes, although DDP treatment resulted in a stronger inducer of both genes. Using two ataxia telangiectasia mutated (ATM) cell lines, the role of ATM in drug-induced p53 phosphorylations was investigated. No differences in drug-induced p53 phosphorylation could be observed, indicating that ATM is not the kinase involved in these phosphorylation events. In addition, inhibition of DNA-dependent protein kinase activity by wortmannin did not abolish p53 phosphorylation at Ser15 and Ser20, again indicating that DNA-PK is unlikely to be the kinase involved. After both taxol and DDP treatments, an activation of hCHK2 was found and this is likely to be responsible for phosphorylation at Ser20. In contrast, only DDP was able to activate ATR, which is the candidate kinase for phosphorylation of Ser15 by this drug. This data clearly suggests that differential mechanisms are involved in phosphorylation and activation of p53 depending on the drug type.
Figures

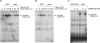


References
-
- KoL J, Prives C. p53: puzzle and paradigm. GenesDev. 1996;56:2649–2654. - PubMed
-
- Levine AJ. p53, the cellular gatekeeper for growth and division. Cell. 1997;88:323–331. - PubMed
-
- Chen X, Ko LJ, Jayaraman L, Prives C. p53 levels, functional domains, and DNA damage determine the extent of the apoptotic response of tumor cells. Genes Dev. 1996;10:2438–2451. - PubMed
-
- Ullrich SJ, Anderson CW, Mercer WE, Appella E. The p53 tumor suppressor protein, a modulator of cell proliferation. J Biol Chem. 1992;267:15259–15262. - PubMed
-
- Wahl AF, Donaldson KL, Fairchild C, Lee FY, Foster SA, Demers GW, Galloway DA. Loss of normal p53 function confers sensitization to Taxol by increasing G2/M arrest and apoptosis. Nat Med. 1996;2:72–79. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
