Rapid cycling of lipid raft markers between the cell surface and Golgi complex
- PMID: 11331304
- PMCID: PMC2190578
- DOI: 10.1083/jcb.153.3.529
Rapid cycling of lipid raft markers between the cell surface and Golgi complex
Abstract
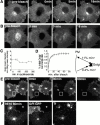
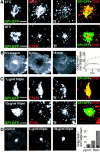
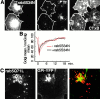
The endocytic itineraries of lipid raft markers, such as glycosyl phosphatidylinositol (GPI)-anchored proteins and glycosphingolipids, are incompletely understood. Here we show that different GPI-anchored proteins have different intracellular distributions; some (such as the folate receptor) accumulate in transferrin-containing compartments, others (such as CD59 and GPI-linked green fluorescent protein [GFP]) accumulate in the Golgi apparatus. Selective photobleaching shows that the Golgi pool of both GPI-GFP and CD59-GFP constantly and rapidly exchanges with the pool of these proteins found on the plasma membrane (PM). We visualized intermediates carrying GPI-GFP from the Golgi apparatus to the PM and separate structures delivering GPI-GFP to the Golgi apparatus.GPI-GFP does not accumulate within endocytic compartments containing transferrin, although it is detected in intracellular structures which are endosomes by the criteria of accessibility to a fluid phase marker and to cholera and shiga toxin B subunits (CTxB and STxB, which are also found in rafts). GPI-GFP and a proportion of the total CTxB and STxB taken up into cells are endocytosed independently of clathrin-associated machinery and are delivered to the Golgi complex via indistinguishable mechanisms. Hence, they enter the Golgi complex in the same intermediates, get there independently of both clathrin and rab5 function, and are excluded from it at 20 degrees C and under conditions of cholesterol sequestration. The PM-Golgi cycling pathway followed by GPI-GFP could serve to regulate lipid raft distribution and function within cells.
Figures





References
-
- Benmerah A., Bayrou M., Cerf-Bensussan N., Dautry-Varsat A. Inhibition of clathrin-coated pit assembly by an Eps15 mutant. J. Cell Sci. 1999;112:1303–1311. - PubMed
-
- Bretscher M.S., Munro S. Cholesterol and the Golgi apparatus. Science. 1993;261:1280–1281. - PubMed
-
- Brown D.A., London E. Functions of lipid rafts in biological membranes. Annu. Rev. Cell Dev. Biol. 1998;14:111–136. - PubMed
-
- Chen H., Fre S., Slepnev V.I., Capua M.R., Takei K., Butler M.H., Di Fiore P.P., De Camilli P. Epsin is an EH-domain-binding protein implicated in clathrin-mediated endocytosis. Nature. 1998;394:793–797. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

