Affinity and location of an internal K+ ion binding site in shaker K channels
- PMID: 11331347
- PMCID: PMC2233657
- DOI: 10.1085/jgp.117.5.373
Affinity and location of an internal K+ ion binding site in shaker K channels
Abstract
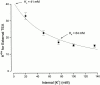
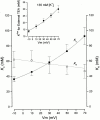
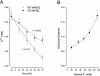
We have examined the interaction between TEA and K+ ions in the pore of Shaker potassium channels. We found that the ability of external TEA to antagonize block of Shaker channels by internal TEA depended on internal K+ ions. In contrast, this antagonism was independent of external K+ concentrations between 0.2 and 40 mM. The external TEA antagonism of internal TEA block increased linearly with the concentration of internal K+ ions. In addition, block by external TEA was significantly enhanced by increases in the internal K+ concentration. These results suggested that external TEA ions do not directly antagonize internal TEA, but rather promote increased occupancy of an internal K+ site by inhibiting the emptying of that site to the external side of the pore. We found this mechanism to be quantitatively consistent with the results and revealed an intrinsic affinity of the site for K+ ions near 65 mM located approximately 7% into the membrane electric field from the internal end of the pore. We also found that the voltage dependence of block by internal TEA was influenced by internal K+ ions. The TEA site (at 0 internal K+) appeared to sense approximately 5% of the field from the internal end of the pore (essentially colocalized with the internal K+ site). These results lead to a refined picture of the number and location of ion binding sites at the inner end of the pore in Shaker K channels.
Figures
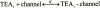
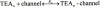
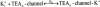
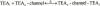
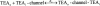




Similar articles
-
Functional identification of ion binding sites at the internal end of the pore in Shaker K+ channels.J Physiol. 2003 May 15;549(Pt 1):107-20. doi: 10.1113/jphysiol.2002.038646. Epub 2003 Mar 28. J Physiol. 2003. PMID: 12665608 Free PMC article.
-
Interaction between quaternary ammonium ions in the pore of potassium channels. Evidence against an electrostatic repulsion mechanism.J Gen Physiol. 2000 Jun;115(6):769-82. doi: 10.1085/jgp.115.6.769. J Gen Physiol. 2000. PMID: 10828250 Free PMC article.
-
External TEA block of shaker K+ channels is coupled to the movement of K+ ions within the selectivity filter.J Gen Physiol. 2003 Aug;122(2):239-46. doi: 10.1085/jgp.200308848. J Gen Physiol. 2003. PMID: 12885878 Free PMC article.
-
Ion channels: a first view of K+ channels in atomic glory.Curr Biol. 1998 Jun 18;8(13):R450-2. doi: 10.1016/s0960-9822(98)70290-8. Curr Biol. 1998. PMID: 9651671 Review.
-
Critical assessment of a proposed model of Shaker.FEBS Lett. 2004 Apr 30;564(3):257-63. doi: 10.1016/S0014-5793(04)00273-X. FEBS Lett. 2004. PMID: 15111106 Review.
Cited by
-
Functional identification of ion binding sites at the internal end of the pore in Shaker K+ channels.J Physiol. 2003 May 15;549(Pt 1):107-20. doi: 10.1113/jphysiol.2002.038646. Epub 2003 Mar 28. J Physiol. 2003. PMID: 12665608 Free PMC article.
-
Block by internal Mg2+ causes voltage-dependent inactivation of Kv1.5.Eur Biophys J. 2006 Dec;36(1):23-34. doi: 10.1007/s00249-006-0085-3. Epub 2006 Aug 11. Eur Biophys J. 2006. PMID: 16902793
-
Na(+) interaction with the pore of Shaker B K(+) channels: zero and low K(+) conditions.J Gen Physiol. 2001 Dec;118(6):639-48. doi: 10.1085/jgp.118.6.639. J Gen Physiol. 2001. PMID: 11723158 Free PMC article.
-
Tail end of the s6 segment: role in permeation in shaker potassium channels.J Gen Physiol. 2002 Jul;120(1):87-97. doi: 10.1085/jgp.20028611. J Gen Physiol. 2002. PMID: 12084778 Free PMC article.
-
Structural insights into neuronal K+ channel-calmodulin complexes.Proc Natl Acad Sci U S A. 2012 Aug 21;109(34):13579-83. doi: 10.1073/pnas.1207606109. Epub 2012 Aug 6. Proc Natl Acad Sci U S A. 2012. PMID: 22869708 Free PMC article.
References
-
- Baukrowitz T., Yellen G. Modulation of K+ current by frequency and external [K+]a tale of two inactivation mechanisms. Neuron. 1995;15:951–960. - PubMed
-
- Baukrowitz T., Yellen G. Use-dependent blockers and exit rate of the last ion from the multi-ion pore of a K+ channel. Science. 1996;271:653–656. - PubMed
-
- Begenisich T., Smith C. Multi-ion nature of potassium channels in squid axons. Curr. Top. Membr. Trans. 1984;22:353–369.
-
- Choi K.L., Mossman C., Aubé C., Yellen G. The internal quaternary ammonium receptor site of Shaker potassium channels. Neuron. 1993;10:533–541. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

