High-fidelity transmission acquired via a developmental decrease in NMDA receptor expression at an auditory synapse
- PMID: 11331363
- PMCID: PMC6762464
- DOI: 10.1523/JNEUROSCI.21-10-03342.2001
High-fidelity transmission acquired via a developmental decrease in NMDA receptor expression at an auditory synapse
Abstract
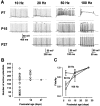
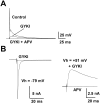
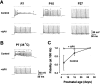
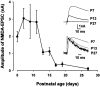
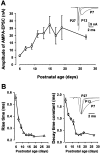
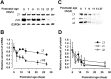
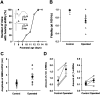
Central auditory relay synapses in mature animals follow high-frequency inputs for computation of sound localization. In immature mice, however, transmission at the calyx of Held synapse in auditory brainstem was inaccurate for high-frequency inputs because the summed slow synaptic potential components caused aberrant firings or blocked action potentials. As the mice matured, synaptic potentials became shorter, with smaller and faster NMDA receptor components, thereby establishing the precise one-to-one transmission for high-frequency inputs. Developmental acquisition of this high-fidelity transmission could be mimicked experimentally in immature mice by blocking NMDA receptors with d(-)2-amino-5-phosphonovaleric acid (d-APV). Furthermore, bilateral cochlear ablations at postnatal day 7 (P7) attenuated the developmental decrease of NMDA receptor expression and prevented the acquisition of high-fidelity transmission. We suggest that auditory activity, which begins at P10-P12 in mice, downregulates the expression of postsynaptic NMDA receptors, thereby contributing to the establishment of high-fidelity synaptic transmission.
Figures
Similar articles
-
Maturation of synaptic transmission at end-bulb synapses of the cochlear nucleus.J Neurosci. 2001 Dec 1;21(23):9487-98. doi: 10.1523/JNEUROSCI.21-23-09487.2001. J Neurosci. 2001. PMID: 11717383 Free PMC article.
-
Fine-tuning an auditory synapse for speed and fidelity: developmental changes in presynaptic waveform, EPSC kinetics, and synaptic plasticity.J Neurosci. 2000 Dec 15;20(24):9162-73. doi: 10.1523/JNEUROSCI.20-24-09162.2000. J Neurosci. 2000. PMID: 11124994 Free PMC article.
-
Receptors underlying excitatory synaptic transmission in slices of the rat anteroventral cochlear nucleus.J Neurophysiol. 1995 Mar;73(3):964-73. doi: 10.1152/jn.1995.73.3.964. J Neurophysiol. 1995. PMID: 7608781
-
Developmental plasticity of NMDA receptors at the calyx of Held synapse.Neuropharmacology. 2021 Sep 15;196:108697. doi: 10.1016/j.neuropharm.2021.108697. Epub 2021 Jul 6. Neuropharmacology. 2021. PMID: 34242682 Review.
-
Contribution of AMPA and NMDA receptors to excitatory responses in the inferior colliculus.Hear Res. 2002 Jun;168(1-2):35-42. doi: 10.1016/s0378-5955(02)00372-6. Hear Res. 2002. PMID: 12117507 Review.
Cited by
-
Linear coding of complex sound spectra by discharge rate in neurons of the medial nucleus of the trapezoid body (MNTB) and its inputs.Front Neural Circuits. 2014 Dec 16;8:144. doi: 10.3389/fncir.2014.00144. eCollection 2014. Front Neural Circuits. 2014. PMID: 25565971 Free PMC article.
-
Synaptogenesis of the calyx of Held: rapid onset of function and one-to-one morphological innervation.J Neurosci. 2006 May 17;26(20):5511-23. doi: 10.1523/JNEUROSCI.5525-05.2006. J Neurosci. 2006. PMID: 16707803 Free PMC article.
-
Maturation of synaptic transmission at end-bulb synapses of the cochlear nucleus.J Neurosci. 2001 Dec 1;21(23):9487-98. doi: 10.1523/JNEUROSCI.21-23-09487.2001. J Neurosci. 2001. PMID: 11717383 Free PMC article.
-
Rapid, activity-dependent plasticity in timing precision in neonatal barrel cortex.J Neurosci. 2006 Apr 19;26(16):4178-87. doi: 10.1523/JNEUROSCI.0150-06.2006. J Neurosci. 2006. PMID: 16624938 Free PMC article.
-
Synaptic plasticity of inhibitory synapses onto medial olivocochlear efferent neurons.J Physiol. 2022 Jun;600(11):2747-2763. doi: 10.1113/JP282815. Epub 2022 May 13. J Physiol. 2022. PMID: 35443073 Free PMC article.
References
-
- Boeckman FA, Aizenman E. Stable transfection of the NR1 subunit in Chinese hamster ovary cells fails to produce a functional N-methyl-d-aspartate receptor. Neurosci Lett. 1994;173:189–192. - PubMed
-
- Carmignoto G, Vicini S. Activity-dependent decrease in NMDA receptor responses during development of the visual cortex. Science. 1992;258:1007–1011. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
