Patched1 interacts with cyclin B1 to regulate cell cycle progression
- PMID: 11331587
- PMCID: PMC125436
- DOI: 10.1093/emboj/20.9.2214
Patched1 interacts with cyclin B1 to regulate cell cycle progression
Abstract
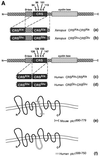
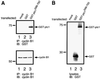
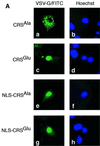
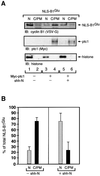
The initiation of mitosis requires the activation of M-phase promoting factor (MPF). MPF activation and its subcellular localization are dependent on the phosphorylation state of its components, cdc2 and cyclin B1. In a two-hybrid screen using a bait protein to mimic phosphorylated cyclin B1, we identified a novel interaction between cyclin B1 and patched1 (ptc1), a tumor suppressor associated with basal cell carcinoma (BCC). Ptc1 interacted specifically with constitutively phosphorylated cyclin B1 derivatives and was able to alter their normal subcellular localization. Furthermore, addition of the ptc1 ligand, sonic hedgehog (shh), disrupts this interaction and allows cyclin B1 to localize to the nucleus. Expression of ptc1 in 293T cells was inhibitory to cell proliferation; this inhibition could be relieved by coexpression of a cyclin B1 derivative that constitutively localizes to the nucleus and that could not interact with ptc1 due to phosphorylation-site mutations to ALA: In addition, we demonstrate that endogenous ptc1 and endogenous cyclin B1 interact in vivo. The findings reported here demonstrate that ptc1 participates in determining the subcellular localization of cyclin B1 and suggest a link between the tumor suppressor activity of ptc1 and the regulation of cell division. Thus, we propose that ptc1 participates in a G(2)/M checkpoint by regulating the localization of MPF.
Figures




References
-
- Borgne A. and Meijer,L. (1996) Sequential dephosphorylation of p34cdc2 on Thr-14 and Tyr-15 at the prophase/metaphase transition. J. Biol. Chem., 271, 27847–27854. - PubMed
-
- Borgne A., Ostvold,A.C., Flament,S. and Meijer,L. (1999) Intra-M phase-promoting factor phosphorylation of cyclin B at the prophase/metaphase transition. J. Biol. Chem., 274, 11977–11986. - PubMed
-
- Chen Y. and Struhl,G. (1996) Dual roles for patched in sequestering and transducing hedgehog. Cell, 87, 553–563. - PubMed
-
- Chidambaram A., Goldstein,A.M., Gailani,M.R., Gerrard,B., Bale,S.J., DiGiovanna,J.J., Bale,A.E. and Dean,M. (1996) Mutations in the human homologue of the drosophila/patched gene in caucasian and African-American nevoid basal cell carcinoma syndrome patients. Cancer Res., 56, 4599–4601. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

