The chaperone GroEL is required for the final assembly of the molybdenum-iron protein of nitrogenase
- PMID: 11331775
- PMCID: PMC33245
- DOI: 10.1073/pnas.101119498
The chaperone GroEL is required for the final assembly of the molybdenum-iron protein of nitrogenase
Abstract
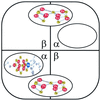
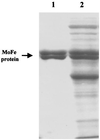
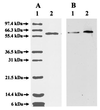
It is known that an E146D site-directed variant of the Azotobacter vinelandii iron protein (Fe protein) is specifically defective in its ability to participate in iron-molybdenum cofactor (FeMoco) insertion. Molybdenum-iron protein (MoFe protein) from the strain expressing the E146D Fe protein is partially ( approximately 45%) FeMoco deficient. The "free" FeMoco that is not inserted accumulates in the cell. We were able to insert this "free" FeMoco into the partially pure FeMoco-deficient MoFe protein. This insertion reaction required crude extract of the DeltanifHDK A. vinelandii strain CA12, Fe protein and MgATP. We used this as an assay to purify a required "insertion" protein. The purified protein was identified as GroEL, based on the molecular mass of its subunit (58.8 kDa), crossreaction with commercially available antibodies raised against E. coli GroEL, and its NH(2)-terminal polypeptide sequence. The NH(2)-terminal polypeptide sequence showed identity of up to 84% to GroEL from various organisms. Purified GroEL of A. vinelandii alone or in combination with MgATP and Fe protein did not support the FeMoco insertion into pure FeMoco-deficient MoFe protein, suggesting that there are still other proteins and/or factors missing. By using GroEL-containing extracts from a DeltanifHDK strain of A. vinelandii CA12 along with FeMoco, Fe protein, and MgATP, we were able to supply all required proteins and/or factors and obtained a fully active reconstituted E146D nifH MoFe protein. The involvement of the molecular chaperone GroEL in the insertion of a metal cluster into an apoprotein may have broad implications for the maturation of other metalloenzymes.
Figures

References
-
- Burgess B K, Lowe D J. Chem Rev. 1996;96:2983–3011. - PubMed
-
- Howard J B, Rees D C. Chem Rev. 1996;96:2965–2982. - PubMed
-
- Howard J B, Rees D C. Annu Rev Biochem. 1994;63:235–264. - PubMed
-
- Seefeldt L C, Dean D R. Acc Chem Res. 1997;30:260–266.
-
- Peters J W, Fischer K, Dean D R. Annu Rev Microbiol. 1995;49:335–366. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

