Biology of E1-deleted adenovirus vectors in nonhuman primate muscle
- PMID: 11333904
- PMCID: PMC114928
- DOI: 10.1128/JVI.75.11.5222-5229.2001
Biology of E1-deleted adenovirus vectors in nonhuman primate muscle
Abstract
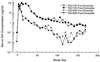
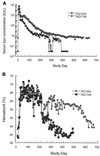
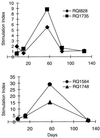
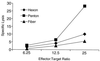
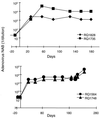
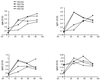
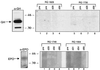
Adenovirus vectors have been studied as vehicles for gene transfer to skeletal muscle, an attractive target for gene therapies for inherited and acquired diseases. In this setting, immune responses to viral proteins and/or transgene products cause inflammation and lead to loss of transgene expression. A few studies in murine models have suggested that the destructive cell-mediated immune response to virally encoded proteins of E1-deleted adenovirus may not contribute to the elimination of transgene-expressing cells. However, the impact of immune responses following intramuscular administration of adenovirus vectors on transgene stability has not been elucidated in larger animal models such as nonhuman primates. Here we demonstrate that intramuscular administration of E1-deleted adenovirus vector expressing rhesus monkey erythropoietin or growth hormone to rhesus monkeys results in generation of a Th1-dependent cytotoxic T-cell response to adenovirus proteins. Transgene expression dropped significantly over time but was still detectable in some animals after 6 months. Systemic levels of adenovirus-specific neutralizing antibodies were generated, which blocked vector readministration. These studies indicate that the cellular and humoral immune response generated to adenovirus proteins, in the context of transgenes encoding self-proteins, hinders long-term transgene expression and readministration with first-generation vectors.
Figures


Similar articles
-
An immunomodulatory procedure that stabilizes transgene expression and permits readministration of E1-deleted adenovirus vectors.Mol Ther. 2001 Mar;3(3):293-301. doi: 10.1006/mthe.2000.0258. Mol Ther. 2001. PMID: 11273770
-
Role of E4 in eliciting CD4 T-cell and B-cell responses to adenovirus vectors delivered to murine and nonhuman primate lungs.J Virol. 1998 Jul;72(7):6138-45. doi: 10.1128/JVI.72.7.6138-6145.1998. J Virol. 1998. PMID: 9621078 Free PMC article.
-
Gene transfer into the liver of nonhuman primates with E1-deleted recombinant adenoviral vectors: safety of readministration.Hum Gene Ther. 1999 Oct 10;10(15):2515-26. doi: 10.1089/10430349950016852. Hum Gene Ther. 1999. PMID: 10543616
-
Immune responses to adenovirus and adeno-associated vectors used for gene therapy of brain diseases: the role of immunological synapses in understanding the cell biology of neuroimmune interactions.Curr Gene Ther. 2007 Oct;7(5):347-60. doi: 10.2174/156652307782151498. Curr Gene Ther. 2007. PMID: 17979681 Free PMC article. Review.
-
Immune response to helper dependent adenoviral mediated liver gene therapy: challenges and prospects.Curr Gene Ther. 2007 Oct;7(5):297-305. doi: 10.2174/156652307782151452. Curr Gene Ther. 2007. PMID: 17979676 Review.
Cited by
-
T cells cause acute immunopathology and are required for long-term survival in mouse adenovirus type 1-induced encephalomyelitis.J Virol. 2003 Sep;77(18):10060-70. doi: 10.1128/jvi.77.18.10060-10070.2003. J Virol. 2003. PMID: 12941916 Free PMC article.
-
Progress and problems when considering gene therapy for GSD-II.Acta Myol. 2007 Jul;26(1):49-52. Acta Myol. 2007. PMID: 17915570 Free PMC article. Review. No abstract available.
-
Clinical and Translational Landscape of Viral Gene Therapies.Cells. 2024 Nov 19;13(22):1916. doi: 10.3390/cells13221916. Cells. 2024. PMID: 39594663 Free PMC article. Review.
-
The road toward AAV-mediated gene therapy of Duchenne muscular dystrophy.Mol Ther. 2025 May 7;33(5):2035-2051. doi: 10.1016/j.ymthe.2025.03.065. Epub 2025 Apr 2. Mol Ther. 2025. PMID: 40181545 Review.
-
Characterization of a permissive epitope insertion site in adenovirus hexon.J Virol. 2006 Jun;80(11):5361-70. doi: 10.1128/JVI.00256-06. J Virol. 2006. PMID: 16699016 Free PMC article.
References
-
- Battershill P E, Clissold S P. Octreotide: a review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential in conditions associated with excessive peptide secretion. Drugs. 1989;38:658–702. - PubMed
-
- Blau H M, Springer M L. Muscle-mediated gene therapy. N Engl J Med. 1995;23:1554–1556. - PubMed
-
- Brody S L, Crystal R G. Adenovirus-mediated in vivo gene transfer. Ann NY Acad Sci. 1994;716:90–103. - PubMed
-
- Bromberg J S, Debruyne L A, Qin L. Interactions between the immune system and gene therapy vectors: bidirectional regulation of response and expression. Adv Immunol. 1998;69:353–409. - PubMed
-
- Chattergoon M, Boyer J, Weiner D B. Genetic immunization: a new era in vaccines and immune therapeutics. FASEB J. 1997;11:753–763. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

