Overexpression of the tobacco Tsi1 gene encoding an EREBP/AP2-type transcription factor enhances resistance against pathogen attack and osmotic stress in tobacco
- PMID: 11340180
- PMCID: PMC135557
- DOI: 10.1105/tpc.13.5.1035
Overexpression of the tobacco Tsi1 gene encoding an EREBP/AP2-type transcription factor enhances resistance against pathogen attack and osmotic stress in tobacco
Abstract
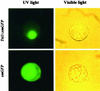
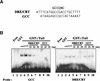
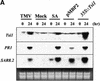
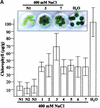
Using mRNA differential display analysis, we isolated a salt-induced transcript that showed a significant sequence homology with an EREBP/AP2 DNA binding motif from oilseed rape plants. With this cDNA fragment as a probe, cDNA clone Tsi1 (for Tobacco stress-induced gene1) was isolated from a tobacco cDNA library. RNA gel blot analysis indicated that transcripts homologous with Tsi1 were induced not only in NaCl-treated leaves but also in leaves treated with ethephon or salicylic acid. Transient expression analysis using a Tsi1::smGFP fusion gene in BY-2 cells indicated that the Tsi1 protein was targeted to the nucleus. Fusion protein of Tsi1 with GAL4 DNA binding domain strongly activated transcription in yeast, and the transactivating activity was localized to the 13 C-terminal amino acids of Tsi1. Electrophoretic mobility shift assays revealed that Tsi1 could bind specifically to the GCC and the DRE/CRT sequences, although the binding activity to the former was stronger than that to the latter. Furthermore, Agrobacterium-mediated transient expression and transgenic plants expressing Tsi1 demonstrated that overexpression of the Tsi1 gene induced expression of several pathogenesis-related genes under normal conditions, resulting in improved tolerance to salt and pathogens. These results suggest that Tsi1 might be involved as a positive trans-acting factor in two separate signal transduction pathways under abiotic and biotic stress.
Figures







References
-
- Abel, S., and Theologis, A. (1994). Transient transformation of Arabidopsis leaf protoplasts: A versatile experimental system to study gene expression. Plant J. 5 421–427. - PubMed
-
- Aono, M., Kubo, A., Saji, H., Tanaka, K., and Kondo, N. (1993). Enhanced tolerance to photooxidative stress of transgenic Nicotiana tabacum with high chloroplastic glutathione reductase activity. Plant Cell Physiol. 34 129–136.
-
- Ausubel, F.M., Brent, R., Kingston, R.E., Moore, D.D., Seidman, J.G., Smith, J.A., and Struhl, K. (1995). Short Protocols in Molecular Biology. (New York: Wiley).
-
- Bol, J.F., Linthorst, H.J.M., and Cornelissen, B.J.C. (1990). Plant pathogenesis-related proteins induced by virus infection. Annu. Rev. Phytopathol. 28 113–138.
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

