Neuralized is essential for a subset of Notch pathway-dependent cell fate decisions during Drosophila eye development
- PMID: 11344304
- PMCID: PMC33265
- DOI: 10.1073/pnas.101135498
Neuralized is essential for a subset of Notch pathway-dependent cell fate decisions during Drosophila eye development
Abstract
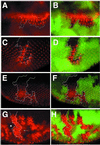
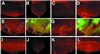
Neuralized (neur) is a neurogenic mutant of Drosophila in which many signaling events mediated by the Notch (N) receptor are disrupted. Here, we analyze the role of neur during eye development. Neur is required in a cell-autonomous fashion to restrict R8 and other photoreceptor fates and is involved in lateral inhibition of interommatidial bristles but is not required for induction of the cone cell fate. The latter contrasts with the absolute requirement for Suppressor of Hairless and the Enhancer of split-Complex for cone cell induction. Using gain-of-function experiments, we further demonstrate that ectopic wild-type and truncated Neur proteins can interfere with multiple N-controlled aspects of eye development, including both neur-dependent and neur-independent processes.
Figures



Similar articles
-
Frizzled/PCP-dependent asymmetric neuralized expression determines R3/R4 fates in the Drosophila eye.Dev Cell. 2006 Dec;11(6):887-94. doi: 10.1016/j.devcel.2006.09.016. Dev Cell. 2006. PMID: 17141162
-
The neuralized homology repeat 1 domain of Drosophila neuralized mediates nuclear envelope association and delta-dependent inhibition of nuclear import.J Mol Biol. 2008 Jan 25;375(4):1125-40. doi: 10.1016/j.jmb.2007.11.043. Epub 2007 Nov 22. J Mol Biol. 2008. PMID: 18076903
-
Two distinct E3 ubiquitin ligases have complementary functions in the regulation of delta and serrate signaling in Drosophila.PLoS Biol. 2005 Apr;3(4):e96. doi: 10.1371/journal.pbio.0030096. Epub 2005 Mar 15. PLoS Biol. 2005. PMID: 15760269 Free PMC article.
-
Eye development: Notch lends a handedness.Curr Biol. 1999 May 20;9(10):R356-60. doi: 10.1016/s0960-9822(99)80226-7. Curr Biol. 1999. PMID: 10339422 Review.
-
Cell Polarity and Notch Signaling: Linked by the E3 Ubiquitin Ligase Neuralized?Bioessays. 2017 Nov;39(11). doi: 10.1002/bies.201700128. Epub 2017 Sep 21. Bioessays. 2017. PMID: 28940548 Review.
Cited by
-
Ethanol hypersensitivity and olfactory discrimination defect in mice lacking a homolog of Drosophila neuralized.Proc Natl Acad Sci U S A. 2001 Aug 14;98(17):9907-12. doi: 10.1073/pnas.171321098. Epub 2001 Jul 31. Proc Natl Acad Sci U S A. 2001. PMID: 11481456 Free PMC article.
-
Origin and evolution of the Notch signalling pathway: an overview from eukaryotic genomes.BMC Evol Biol. 2009 Oct 13;9:249. doi: 10.1186/1471-2148-9-249. BMC Evol Biol. 2009. PMID: 19825158 Free PMC article.
-
MicroRNA targets in Drosophila.Genome Biol. 2003;5(1):R1. doi: 10.1186/gb-2003-5-1-r1. Epub 2003 Dec 12. Genome Biol. 2003. PMID: 14709173 Free PMC article.
-
A Notch updated.J Cell Biol. 2009 Mar 9;184(5):621-9. doi: 10.1083/jcb.200811141. Epub 2009 Mar 2. J Cell Biol. 2009. PMID: 19255248 Free PMC article. Review.
-
Drosophila EHBP1 regulates Scabrous secretion during Notch-mediated lateral inhibition.J Cell Sci. 2013 Aug 15;126(Pt 16):3686-96. doi: 10.1242/jcs.126292. Epub 2013 Jun 20. J Cell Sci. 2013. PMID: 23788431 Free PMC article.
References
-
- Cagan R L, Ready D F. Genes Dev. 1989;3:1099–1112. - PubMed
-
- Go M J, Eastman D S, Artavanis-Tsakonas S. Development (Cambridge, UK) 1998;125:2031–2040. - PubMed
-
- Papayannopoulos V, Tomlinson A, Panin V M, Rauskolb C, Irvine K D. Science. 1998;281:2031–2034. - PubMed
-
- Dominguez M, de Celis J F. Nature (London) 1998;396:276–278. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

