Knockout of the rodent malaria parasite chitinase pbCHT1 reduces infectivity to mosquitoes
- PMID: 11349074
- PMCID: PMC98467
- DOI: 10.1128/IAI.69.6.4041-4047.2001
Knockout of the rodent malaria parasite chitinase pbCHT1 reduces infectivity to mosquitoes
Abstract
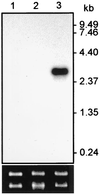
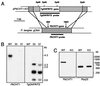
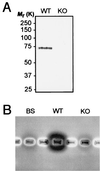
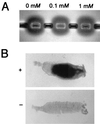
During mosquito transmission, malaria ookinetes must cross a chitin-containing structure known as the peritrophic matrix (PM), which surrounds the infected blood meal in the mosquito midgut. In turn, ookinetes produce multiple chitinase activities presumably aimed at disrupting this physical barrier to allow ookinete invasion of the midgut epithelium. Plasmodium chitinase activities are demonstrated targets for human and avian malaria transmission blockade with the chitinase inhibitor allosamidin. Here, we identify and characterize the first chitinase gene of a rodent malaria parasite, Plasmodium berghei. We show that the gene, named PbCHT1, is a structural ortholog of PgCHT1 of the avian malaria parasite Plasmodium gallinaceum and a paralog of PfCHT1 of the human malaria parasite Plasmodium falciparum. Targeted disruption of PbCHT1 reduced parasite infectivity in Anopheles stephensi mosquitoes by up to 90%. Reductions in infectivity were also observed in ookinete feeds-an artificial situation where midgut invasion occurs before PM formation-suggesting that PbCHT1 plays a role other than PM disruption. PbCHT1 null mutants had no residual ookinete-derived chitinase activity in vitro, suggesting that P. berghei ookinetes express only one chitinase gene. Moreover, PbCHT1 activity appeared insensitive to allosamidin inhibition, an observation that raises questions about the use of allosamidin and components like it as potential malaria transmission-blocking drugs. Taken together, these findings suggest a fundamental divergence among rodent, avian, and human malaria parasite chitinases, with implications for the evolution of Plasmodium-mosquito interactions.
Figures

Similar articles
-
Plasmodium chitinases: revisiting a target of transmission-blockade against malaria.Protein Sci. 2021 Aug;30(8):1493-1501. doi: 10.1002/pro.4095. Epub 2021 May 8. Protein Sci. 2021. PMID: 33934433 Free PMC article. Review.
-
A Hetero-Multimeric Chitinase-Containing Plasmodium falciparum and Plasmodium gallinaceum Ookinete-Secreted Protein Complex Involved in Mosquito Midgut Invasion.Front Cell Infect Microbiol. 2021 Jan 8;10:615343. doi: 10.3389/fcimb.2020.615343. eCollection 2020. Front Cell Infect Microbiol. 2021. PMID: 33489941 Free PMC article.
-
The chitinase PfCHT1 from the human malaria parasite Plasmodium falciparum lacks proenzyme and chitin-binding domains and displays unique substrate preferences.Proc Natl Acad Sci U S A. 1999 Nov 23;96(24):14061-6. doi: 10.1073/pnas.96.24.14061. Proc Natl Acad Sci U S A. 1999. PMID: 10570198 Free PMC article.
-
Chitinases of the avian malaria parasite Plasmodium gallinaceum, a class of enzymes necessary for parasite invasion of the mosquito midgut.J Biol Chem. 2000 Apr 7;275(14):10331-41. doi: 10.1074/jbc.275.14.10331. J Biol Chem. 2000. PMID: 10744721
-
Plasmodium ookinete-secreted chitinase and parasite penetration of the mosquito peritrophic matrix.Trends Parasitol. 2001 Jun;17(6):269-72. doi: 10.1016/s1471-4922(01)01918-3. Trends Parasitol. 2001. PMID: 11378031 Review.
Cited by
-
Molecular identification of the chitinase genes in Plasmodium relictum.Malar J. 2014 Jun 18;13:239. doi: 10.1186/1475-2875-13-239. Malar J. 2014. PMID: 24943514 Free PMC article.
-
Reverse genetics screen identifies six proteins important for malaria development in the mosquito.Mol Microbiol. 2008 Oct;70(1):209-20. doi: 10.1111/j.1365-2958.2008.06407.x. Epub 2008 Aug 27. Mol Microbiol. 2008. PMID: 18761621 Free PMC article.
-
Microbiota-induced peritrophic matrix regulates midgut homeostasis and prevents systemic infection of malaria vector mosquitoes.PLoS Pathog. 2017 May 17;13(5):e1006391. doi: 10.1371/journal.ppat.1006391. eCollection 2017 May. PLoS Pathog. 2017. PMID: 28545061 Free PMC article.
-
Plasmodium chitinases: revisiting a target of transmission-blockade against malaria.Protein Sci. 2021 Aug;30(8):1493-1501. doi: 10.1002/pro.4095. Epub 2021 May 8. Protein Sci. 2021. PMID: 33934433 Free PMC article. Review.
-
Enzymatic characterization of the Plasmodium vivax chitinase, a potential malaria transmission-blocking target.Parasitol Int. 2009 Sep;58(3):243-8. doi: 10.1016/j.parint.2009.05.002. Epub 2009 May 8. Parasitol Int. 2009. PMID: 19427918 Free PMC article.
References
-
- Berner R, Rudin W, Hecker H. Peritrophic membranes and protease activity in the midgut of the malaria mosquito, Anopheles stephensi (Liston) (Insecta: Diptera), under normal and experimental conditions. J Ultrastruct Res. 1983;83:195–204. - PubMed
-
- Claudianos C, Russell R J, Oakeshott J G. The same amino acid substitution in orthologous esterases confers organophosphate resistance on the house fly and a blowfly. Insect Biochem Mol Biol. 1999;29:675–686. - PubMed
-
- Dessens J T, Margos G, Rodriguez M C, Sinden R E. Identification of differentially regulated genes of Plasmodium by suppression subtractive hybridization. Parasitol Today. 2000;16:354–356. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources

