Coiled-coil domain of enteropathogenic Escherichia coli type III secreted protein EspD is involved in EspA filament-mediated cell attachment and hemolysis
- PMID: 11349076
- PMCID: PMC98469
- DOI: 10.1128/IAI.69.6.4055-4064.2001
Coiled-coil domain of enteropathogenic Escherichia coli type III secreted protein EspD is involved in EspA filament-mediated cell attachment and hemolysis
Abstract
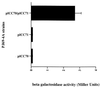
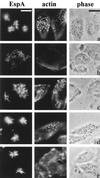
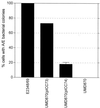
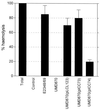
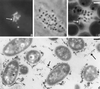
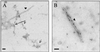
Many animal and plant pathogens use type III secretion systems to secrete key virulence factors, some directly into the host cell cytosol. However, the basis for such protein translocation has yet to be fully elucidated for any type III secretion system. We have previously shown that in enteropathogenic and enterohemorrhagic Escherichia coli the type III secreted protein EspA is assembled into a filamentous organelle that attaches the bacterium to the plasma membrane of the host cell. Formation of EspA filaments is dependent on expression of another type III secreted protein, EspD. The carboxy terminus of EspD, a protein involved in formation of the translocation pore in the host cell membrane, is predicted to adopt a coiled-coil conformation with 99% probability. Here, we demonstrate EspD-EspD protein interaction using the yeast two-hybrid system and column overlays. Nonconservative triple amino acid substitutions of specific EspD carboxy-terminal residues generated an enteropathogenic E. coli mutant that was attenuated in its ability to induce attaching and effacing lesions on HEp-2 cells. Although the mutation had no effect on EspA filament biosynthesis, it also resulted in reduced binding to and reduced hemolysis of red blood cells. These results segregate, for the first time, functional domains of EspD that control EspA filament length from EspD-mediated cell attachment and pore formation.
Figures




Similar articles
-
Supermolecular structure of the enteropathogenic Escherichia coli type III secretion system and its direct interaction with the EspA-sheath-like structure.Proc Natl Acad Sci U S A. 2001 Sep 25;98(20):11638-43. doi: 10.1073/pnas.191378598. Epub 2001 Sep 18. Proc Natl Acad Sci U S A. 2001. PMID: 11562461 Free PMC article.
-
Alpha 1-antitrypsin binds to and interferes with functionality of EspB from atypical and typical enteropathogenic Escherichia coli strains.Infect Immun. 2004 Aug;72(8):4344-50. doi: 10.1128/IAI.72.8.4344-4350.2004. Infect Immun. 2004. PMID: 15271889 Free PMC article.
-
EspA filament-mediated protein translocation into red blood cells.Cell Microbiol. 2001 Apr;3(4):213-22. doi: 10.1046/j.1462-5822.2001.00105.x. Cell Microbiol. 2001. PMID: 11298645
-
Enteropathogenic Escherichia coli: cellular harassment.Curr Opin Microbiol. 1999 Feb;2(1):83-8. doi: 10.1016/s1369-5274(99)80014-9. Curr Opin Microbiol. 1999. PMID: 10047555 Review.
-
Intimate interactions of enteropathogenic Escherichia coli at the host cell surface.Curr Opin Infect Dis. 2001 Oct;14(5):559-65. doi: 10.1097/00001432-200110000-00009. Curr Opin Infect Dis. 2001. PMID: 11964876 Review.
Cited by
-
Bacterial outer membrane vesicles provide an alternative pathway for trafficking of Escherichia coli O157 type III secreted effectors to epithelial cells.mSphere. 2023 Dec 20;8(6):e0052023. doi: 10.1128/msphere.00520-23. Epub 2023 Nov 6. mSphere. 2023. PMID: 37929984 Free PMC article.
-
A type VII secretion system in Group B Streptococcus mediates cytotoxicity and virulence.PLoS Pathog. 2021 Dec 6;17(12):e1010121. doi: 10.1371/journal.ppat.1010121. eCollection 2021 Dec. PLoS Pathog. 2021. PMID: 34871327 Free PMC article.
-
Detection of secretory immunoglobulin A in human colostrum as mucosal immune response against proteins of the type III secretion system of Salmonella, Shigella and enteropathogenic Escherichia coli.Pediatr Infect Dis J. 2013 Oct;32(10):1122-6. doi: 10.1097/INF.0b013e318293306c. Pediatr Infect Dis J. 2013. PMID: 23538526 Free PMC article.
-
Polarity of enteropathogenic Escherichia coli EspA filament assembly and protein secretion.J Bacteriol. 2005 Apr;187(8):2881-9. doi: 10.1128/JB.187.8.2881-2889.2005. J Bacteriol. 2005. PMID: 15805534 Free PMC article.
-
The Type III Secretion System (T3SS)-Translocon of Atypical Enteropathogenic Escherichia coli (aEPEC) Can Mediate Adherence.Front Microbiol. 2019 Jul 9;10:1527. doi: 10.3389/fmicb.2019.01527. eCollection 2019. Front Microbiol. 2019. PMID: 31338081 Free PMC article.
References
-
- Cornelis G, Van Gijsegem F. Assembly and function of type III secretory systems. Annu Rev Microbiol. 2000;54:735–774. - PubMed
-
- Cravioto A, Gross R J, Scotland S M, Rowe B. An adhesive factor found in strains of Escherichia coli belonging to the traditional infantile enteropathogenic serogroups. Curr Microbiol. 1979;3:95–99.
-
- Deibel C, Kramer S, Chakraborty T, Ebel F. EspE, a novel secreted protein of attaching and effacing bacteria, is directly translocated into infected host cells, where it appears as a tyrosine-phosphorylated 90 kDa protein. Mol Microbiol. 1998;28:463–474. - PubMed
-
- Delahay R M, Knutton S, Shaw R K, Hartland E L, Pallen M J, Frankel G. The coiled-coil domain of EspA is essential for the assembly of the type III secretion translocon on the surface of enteropathogenic E. coli. J Biol Chem. 1999;274:35969–35974. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

