Activation of alpha(v)beta3-vitronectin binding is a multistage process in which increases in bond strength are dependent on Y747 and Y759 in the cytoplasmic domain of beta3
- PMID: 11359918
- PMCID: PMC34580
- DOI: 10.1091/mbc.12.5.1227
Activation of alpha(v)beta3-vitronectin binding is a multistage process in which increases in bond strength are dependent on Y747 and Y759 in the cytoplasmic domain of beta3
Abstract
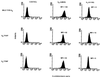
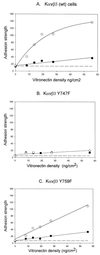
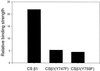
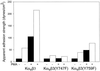
Integrin receptors serve as mechanical links between the cell and its structural environment. Using alpha(v)beta3 integrin expressed in K562 cells as a model system, the process by which the mechanical connection between alpha(v)beta3 and vitronectin develops was analyzed by measuring the resistance of these bonds to mechanical separation. Three distinct stages of activation, as defined by increases in the alpha(v)beta3-vitronectin binding strength, were defined by mutational, biochemical, and biomechanical analyses. Activation to the low binding strength stage 1 occurs through interaction with the vitronectin ligand and leads to the phosphorylation of Y747 in the beta3 subunit. Stage 2 is characterized by a 4-fold increase in binding strength and is dependent on stage1 and the phosphorylation of Y747. Stage 3 is characterized by a further 2.5-fold increase in binding strength and is dependent on stage 2 events and the availability of Y759 for interaction with cellular proteins. The Y747F mutant blocked the transition from stage 1 to stage 2, and the Y759F blocked the transition from stage 2 to stage 3. The data suggest a model for tension-induced activation of alpha(v)beta3 integrin.
Figures







References
-
- Bazzoni G, Hemler ME. Are changes in integrin affinity and conformation overemphasized? Trends Biochem Sci. 1998;23:30–34. - PubMed
-
- Bennett JS, Zigmond S, Vilaire G, Cunningham ME, Bednar B. The platelet cytoskeleton regulates the affinity of the integrin alpha(IIb)beta(3) for fibrinogen. J Biol Chem. 1999;274:25301–25307. - PubMed
-
- Blystone SD, Lindberg FP, Williams MP, McHugh KP, Brown EJ. Inducible tyrosine phosphorylation of the beta3 integrin requires the alphaV integrin cytoplasmic tail. J Biol Chem. 1996;271:31458–31462. - PubMed
-
- Blystone SD, Williams MP, Slater SE, Brown EJ. Requirement of integrin beta3 tyrosine 747 for beta3 tyrosine phosphorylation and regulation of alphavbeta3 avidity. J Biol Chem. 1997;272:28757–28761. - PubMed
-
- Boettiger D, George-Weinstein M, Menko AS. Triggering terminal myogenic differentiation. In: Stockdale F, Kedes L, editors. UCLA Symposia on Molecular and Cellular Biology, New Series: Cellular and Molecular Biology of Muscle Development. New York: Alan R. Liss, Inc.; 1989. pp. 57–66.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

