Phosphorylation and microtubule association of the Opitz syndrome protein mid-1 is regulated by protein phosphatase 2A via binding to the regulatory subunit alpha 4
- PMID: 11371618
- PMCID: PMC34408
- DOI: 10.1073/pnas.111154698
Phosphorylation and microtubule association of the Opitz syndrome protein mid-1 is regulated by protein phosphatase 2A via binding to the regulatory subunit alpha 4
Abstract
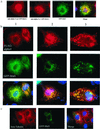
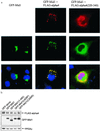
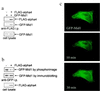
Opitz syndrome (OS) is a human genetic disease characterized by deformities such as cleft palate that are attributable to defects in embryonic development at the midline. Gene mapping has identified OS mutations within a protein called Mid1. Wild-type Mid1 predominantly colocalizes with microtubules, in contrast to mutant versions of Mid1 that appear clustered in the cytosol. Using yeast two-hybrid screening, we found that the alpha4-subunit of protein phosphatases 2A/4/6 binds Mid1. Epitope-tagged alpha4 coimmunoprecipitated endogenous or coexpressed Mid1 from COS7 cells, and this required only the conserved C-terminal region of alpha4. Localization of Mid1 and alpha4 was influenced by one another in transiently transfected cells. Mid1 could recruit alpha4 onto microtubules, and high levels of alpha4 could displace Mid1 into the cytosol. Metabolic (32)P labeling of cells showed that Mid1 is a phosphoprotein, and coexpression of full-length alpha4 decreased Mid1 phosphorylation, indicative of a functional interaction. Association of green fluorescent protein-Mid1 with microtubules in living cells was perturbed by inhibitors of MAP kinase activation. The conclusion is that Mid1 association with microtubules, which seems important for normal midline development, is regulated by dynamic phosphorylation involving MAP kinase and protein phosphatase that is targeted specifically to Mid1 by alpha4. Human birth defects may result from environmental or genetic disruption of this regulatory cycle.
Figures

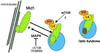
Similar articles
-
Active transport of the ubiquitin ligase MID1 along the microtubules is regulated by protein phosphatase 2A.PLoS One. 2008;3(10):e3507. doi: 10.1371/journal.pone.0003507. Epub 2008 Oct 24. PLoS One. 2008. PMID: 18949047 Free PMC article.
-
Mig12, a novel Opitz syndrome gene product partner, is expressed in the embryonic ventral midline and co-operates with Mid1 to bundle and stabilize microtubules.BMC Cell Biol. 2004 Feb 29;5:9. doi: 10.1186/1471-2121-5-9. BMC Cell Biol. 2004. PMID: 15070402 Free PMC article.
-
MID1, mutated in Opitz syndrome, encodes an ubiquitin ligase that targets phosphatase 2A for degradation.Nat Genet. 2001 Nov;29(3):287-94. doi: 10.1038/ng762. Nat Genet. 2001. PMID: 11685209
-
The MID1/PP2A complex: a key to the pathogenesis of Opitz BBB/G syndrome.Bioessays. 2003 Apr;25(4):356-66. doi: 10.1002/bies.10256. Bioessays. 2003. PMID: 12655643 Review.
-
The MID1 gene product in physiology and disease.Gene. 2020 Jul 15;747:144655. doi: 10.1016/j.gene.2020.144655. Epub 2020 Apr 10. Gene. 2020. PMID: 32283114 Free PMC article. Review.
Cited by
-
Lack of Mid1, the mouse ortholog of the Opitz syndrome gene, causes abnormal development of the anterior cerebellar vermis.J Neurosci. 2010 Feb 24;30(8):2880-7. doi: 10.1523/JNEUROSCI.4196-09.2010. J Neurosci. 2010. PMID: 20181585 Free PMC article.
-
Microcystin-LR (MCLR) induces a compensation of PP2A activity mediated by α4 protein in HEK293 cells.Int J Biol Sci. 2011;7(6):740-52. doi: 10.7150/ijbs.7.740. Epub 2011 Jun 8. Int J Biol Sci. 2011. PMID: 21698000 Free PMC article.
-
MID1 catalyzes the ubiquitination of protein phosphatase 2A and mutations within its Bbox1 domain disrupt polyubiquitination of alpha4 but not of PP2Ac.PLoS One. 2014 Sep 10;9(9):e107428. doi: 10.1371/journal.pone.0107428. eCollection 2014. PLoS One. 2014. PMID: 25207814 Free PMC article.
-
NMR studies of the C-terminus of alpha4 reveal possible mechanism of its interaction with MID1 and protein phosphatase 2A.PLoS One. 2011;6(12):e28877. doi: 10.1371/journal.pone.0028877. Epub 2011 Dec 14. PLoS One. 2011. PMID: 22194938 Free PMC article.
-
Cytokine activation of p38 mitogen-activated protein kinase and apoptosis is opposed by alpha-4 targeting of protein phosphatase 2A for site-specific dephosphorylation of MEK3.Mol Cell Biol. 2007 Jun;27(12):4217-27. doi: 10.1128/MCB.00067-07. Epub 2007 Apr 16. Mol Cell Biol. 2007. PMID: 17438131 Free PMC article.
References
-
- Quaderi N A, Schweiger S, Gaudenz K, Franco B, Rugarli E I, Berger W, Feldman G J, Volta M, Andolfi G, Gilgenkrantz S, et al. Nat Genet. 1997;17:285–291. - PubMed
-
- Cox T C, Allen L R, Cox L L, Hopwood B, Goodwin B, Haan E, Suthers G K. Hum Mol Genet. 2000;9:2553–2562. - PubMed
-
- Dal Zotto L, Quaderi N A, Elliott R, Lingerfelter P A, Carrel L, Valsecchi V, Montini E, Yen C H, Chapman V, Kalcheva I, et al. Hum Mol Genet. 1998;7:489–499. - PubMed
-
- Cainarca S, Messali S, Ballabio A, Meroni G. Hum Mol Genet. 1999;8:1387–1396. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

