Proinflammatory cytokines regulate human glucocorticoid receptor gene expression and lead to the accumulation of the dominant negative beta isoform: a mechanism for the generation of glucocorticoid resistance
- PMID: 11381138
- PMCID: PMC34444
- DOI: 10.1073/pnas.121455098
Proinflammatory cytokines regulate human glucocorticoid receptor gene expression and lead to the accumulation of the dominant negative beta isoform: a mechanism for the generation of glucocorticoid resistance
Abstract
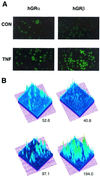
Inflammatory responses in many cell types are coordinately regulated by the opposing actions of NF-kappaB and the glucocorticoid receptor (GR). The human glucocorticoid receptor (hGR) gene encodes two protein isoforms: a cytoplasmic alpha form (GRalpha), which binds hormone, translocates to the nucleus, and regulates gene transcription, and a nuclear localized beta isoform (GRbeta), which does not bind known ligands and attenuates GRalpha action. We report here the identification of a tumor necrosis factor (TNF)-responsive NF-kappaB DNA binding site 5' to the hGR promoter that leads to a 1.5-fold increase in GRalpha mRNA and a 2.0-fold increase in GRbeta mRNA in HeLaS3 cells, which endogenously express both GR isoforms. However, TNF-alpha treatment disproportionately increased the steady-state levels of the GRbeta protein isoform over GRalpha, making GRbeta the predominant endogenous receptor isoform. Similar results were observed following treatment of human CEMC7 lymphoid cells with TNF-alpha or IL-1. The increase in GRbeta protein expression correlated with the development of glucocorticoid resistance.
Figures






Similar articles
-
Overexpression of the human glucocorticoid receptor alpha and beta isoforms inhibits AP-1 and NF-kappaB activities hormone independently.J Mol Med (Berl). 2002 May;80(5):309-18. doi: 10.1007/s00109-001-0302-6. Epub 2001 Dec 7. J Mol Med (Berl). 2002. PMID: 12021843
-
Differential mRNA expression of glucocorticoid receptor alpha and beta is associated with glucocorticoid sensitivity of acute lymphoblastic leukemia in children.Pediatr Blood Cancer. 2005 Aug;45(2):121-7. doi: 10.1002/pbc.20308. Pediatr Blood Cancer. 2005. PMID: 15704223
-
Interindividual glucocorticoid sensitivity in young healthy subjects: the role of glucocorticoid receptor alpha and beta isoforms ratio.Horm Metab Res. 2007 Jun;39(6):425-9. doi: 10.1055/s-2007-980191. Horm Metab Res. 2007. PMID: 17578759
-
Cortisol resistance in conditions such as asthma and the involvement of 11beta-HSD-2: a hypothesis.Horm Metab Res. 2006 Jun;38(6):368-76. doi: 10.1055/s-2006-944530. Horm Metab Res. 2006. PMID: 16823718 Review.
-
Glucocorticoid receptor in human respiratory epithelial cells.Neuroimmunomodulation. 2009;16(5):290-9. doi: 10.1159/000216187. Epub 2009 Jun 29. Neuroimmunomodulation. 2009. PMID: 19571590 Review.
Cited by
-
Deconstructing the roles of glucocorticoids in adipose tissue biology and the development of central obesity.Biochim Biophys Acta. 2014 Mar;1842(3):473-81. doi: 10.1016/j.bbadis.2013.05.029. Epub 2013 Jun 2. Biochim Biophys Acta. 2014. PMID: 23735216 Free PMC article. Review.
-
A novel BAT3 sequence generated by alternative RNA splicing of exon 11B displays cell type-specific expression and impacts on subcellular localization.PLoS One. 2012;7(4):e35972. doi: 10.1371/journal.pone.0035972. Epub 2012 Apr 25. PLoS One. 2012. PMID: 22558287 Free PMC article.
-
Bronchoprotection and bronchorelaxation in asthma: New targets, and new ways to target the old ones.Pharmacol Ther. 2016 Aug;164:82-96. doi: 10.1016/j.pharmthera.2016.04.002. Epub 2016 Apr 23. Pharmacol Ther. 2016. PMID: 27113408 Free PMC article. Review.
-
[Association of the structure of the glucocorticoid receptor and single nucleotide NR3C1 gene polymorphisms with metabolic disorders].Probl Endokrinol (Mosk). 2023 Feb 25;69(1):50-58. doi: 10.14341/probl13160. Probl Endokrinol (Mosk). 2023. PMID: 36842077 Free PMC article. Review. Russian.
-
Could GILZ Be the Answer to Glucocorticoid Toxicity in Lupus?Front Immunol. 2019 Jul 17;10:1684. doi: 10.3389/fimmu.2019.01684. eCollection 2019. Front Immunol. 2019. PMID: 31379872 Free PMC article. Review.
References
-
- Baeuerle P A. Curr Biol. 1998;8:R19–R22. - PubMed
-
- Baldwin A S. Annu Rev Immunol. 1996;14:649–681. - PubMed
-
- Sen R, Baltimore D. Cell. 1986;46:706–716.
-
- Caldenhoven E, Liden J, Wissink S, Van de Stolpe A, Raaijimakers J, Koenderman L, Okret S, Gustafsson J A, Van der Saag P T. Mol Endocrinol. 1995;9:401–412. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

