Activated MEK5 induces serial assembly of sarcomeres and eccentric cardiac hypertrophy
- PMID: 11387209
- PMCID: PMC125475
- DOI: 10.1093/emboj/20.11.2757
Activated MEK5 induces serial assembly of sarcomeres and eccentric cardiac hypertrophy
Abstract
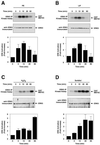
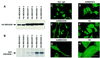
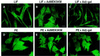
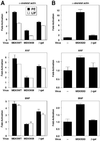
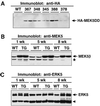
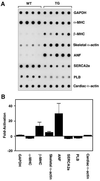
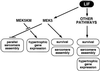
Mitogen-activated protein kinase (MAPK) pathways couple intrinsic and extrinsic signals to hypertrophic growth of cardiomyocytes. The MAPK kinase MEK5 activates the MAPK ERK5. To investigate the potential involvement of MEK5-ERK5 in cardiac hypertrophy, we expressed constitutively active and dominant-negative forms of MEK5 in cardiomyocytes in vitro. MEK5 induced a form of hypertrophy in which cardiomyocytes acquired an elongated morphology and sarcomeres were assembled in a serial manner. The cytokine leukemia inhibitory factor (LIF), which stimulates MEK5 activity, evoked a similar response. Moreover, a dominant-negative MEK5 mutant specifically blocked LIF-induced elongation of cardiomyocytes and reduced expression of fetal cardiac genes without blocking other aspects of LIF-induced hypertrophy. Consistent with the ability of MEK5 to induce serial assembly of sarcomeres in vitro, cardiac-specific expression of activated MEK5 in transgenic mice resulted in eccentric cardiac hypertrophy that progressed to dilated cardiomyopathy and sudden death. These findings reveal a specific role for MEK5-ERK5 in the induction of eccentric cardiac hypertrophy and in transduction of cytokine signals that regulate serial sarcomere assembly.
Figures



Similar articles
-
MEKK2 associates with the adapter protein Lad/RIBP and regulates the MEK5-BMK1/ERK5 pathway.J Biol Chem. 2001 Feb 16;276(7):5093-100. doi: 10.1074/jbc.M003719200. Epub 2000 Nov 9. J Biol Chem. 2001. PMID: 11073940
-
Activation of gp130 transduces hypertrophic signal through interaction of scaffolding/docking protein Gab1 with tyrosine phosphatase SHP2 in cardiomyocytes.Circ Res. 2003 Aug 8;93(3):221-9. doi: 10.1161/01.RES.0000085562.48906.4A. Epub 2003 Jul 10. Circ Res. 2003. PMID: 12855672
-
Specific role of the extracellular signal-regulated kinase pathway in angiotensin II-induced cardiac hypertrophy in vitro.Biochem J. 2000 Apr 1;347 Pt 1(Pt 1):275-84. Biochem J. 2000. PMID: 10727428 Free PMC article.
-
Calcineurin-NFAT signaling regulates the cardiac hypertrophic response in coordination with the MAPKs.Cardiovasc Res. 2004 Aug 15;63(3):467-75. doi: 10.1016/j.cardiores.2004.01.021. Cardiovasc Res. 2004. PMID: 15276472 Review.
-
The MEK5/ERK5 signalling pathway in cancer: a promising novel therapeutic target.Drug Discov Today. 2016 Oct;21(10):1654-1663. doi: 10.1016/j.drudis.2016.06.010. Epub 2016 Jun 16. Drug Discov Today. 2016. PMID: 27320690 Review.
Cited by
-
RSK3: A regulator of pathological cardiac remodeling.IUBMB Life. 2015 May;67(5):331-7. doi: 10.1002/iub.1383. Epub 2015 May 19. IUBMB Life. 2015. PMID: 25988524 Free PMC article. Review.
-
Re-employment of developmental transcription factors in adult heart disease.Semin Cell Dev Biol. 2007 Feb;18(1):117-31. doi: 10.1016/j.semcdb.2006.11.012. Epub 2006 Nov 24. Semin Cell Dev Biol. 2007. PMID: 17161634 Free PMC article. Review.
-
Caenorhabditis elegans and its applicability to studies on restless legs syndrome.Adv Pharmacol. 2019;84:147-174. doi: 10.1016/bs.apha.2018.12.003. Epub 2019 Mar 14. Adv Pharmacol. 2019. PMID: 31229169 Free PMC article. Review.
-
USP20 deletion promotes eccentric cardiac remodeling in response to pressure overload and increases mortality.Am J Physiol Heart Circ Physiol. 2024 Nov 1;327(5):H1257-H1271. doi: 10.1152/ajpheart.00329.2024. Epub 2024 Oct 4. Am J Physiol Heart Circ Physiol. 2024. PMID: 39365672
-
mAKAP compartmentalizes oxygen-dependent control of HIF-1alpha.Sci Signal. 2008 Dec 23;1(51):ra18. doi: 10.1126/scisignal.2000026. Sci Signal. 2008. PMID: 19109240 Free PMC article.
References
-
- Abe J., Kusuhara,M., Ulevitch,R.J., Berk,B.C. and Lee,J.D. (1996) Big mitogen-activated protein kinase 1 (BMK1) is a redox-sensitive kinase. J. Biol. Chem., 271, 16586–16590. - PubMed
-
- Abe J., Baines,C.P. and Berk,B.C. (2000) Role of mitogen-activated protein kinases in ischemia and reperfusion injury: the good and the bad. Circ. Res., 86, 607–609. - PubMed
-
- Becker T.C., Noel,R.J., Coats,W.S., Gomez-Foix,A.M., Alam,T., Gerard,R.D. and Newgard,C.B. (1994) Use of recombinant adenovirus for metabolic engineering of mammalian cells. Methods Cell Biol., 43, 161–189. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

