Tumorigenic potential of extracellular matrix metalloproteinase inducer
- PMID: 11395366
- PMCID: PMC1891983
- DOI: 10.1016/S0002-9440(10)64660-3
Tumorigenic potential of extracellular matrix metalloproteinase inducer
Abstract
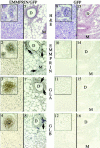
Extracellular matrix metalloproteinase inducer (EMMPRIN), a glycoprotein present on the cancer cell plasma membrane, enhances fibroblast synthesis of matrix metalloproteinases (MMPs). The demonstration that peritumoral fibroblasts synthesize most of the MMPs in human tumors rather than the cancer cells themselves has ignited interest in the role of EMMPRIN in tumor dissemination. In this report we have demonstrated a role for EMMPRIN in cancer progression. Human MDA-MB-436 breast cancer cells, which are tumorigenic but slow growing in vivo, were transfected with EMMPRIN cDNA and injected orthotopically into mammary tissue of female NCr nu/nu mice. Green fluorescent protein was used to visualize metastases. In three experiments, breast cancer cell clones transfected with EMMPRIN cDNA were considerably more tumorigenic and invasive than plasmid-transfected cancer cells. Increased gelatinase A and gelatinase B expression (demonstrated by in situ hybridization and gelatin substrate zymography) was demonstrated in EMMPRIN-enhanced tumors. In contrast to de novo breast cancers in humans, human tumors transplanted into mice elicited minimal stromal or inflammatory cell reactions. Based on these experimental studies and our previous demonstration that EMMPRIN is prominently displayed in human cancer tissue, we propose that EMMPRIN plays an important role in cancer progression by increasing synthesis of MMPs.
Figures


References
-
- Biswas C, Zhang Y, DeCastro R, Guuo H, Nakamura T, Kataoka H, Nabeshima K: The human tumor cell-derived collagenase stimulatory factor (renamed EMMPRIN) is a member of the immunoglobulin superfamily. Cancer Res 1995, 55:434-439 - PubMed
-
- Kataoka H, DeCastro R, Zucker S, Biswas C: The tumor cell-derived Collagenase Stimulating Factor, TCSP, increases expression of interstitial collagenase, stromelysin and 72 kDa gelatinase. Cancer Res 1993, 53:3155-3158 - PubMed
-
- Guo H, Zucker S, Gordon M, Toole BP, Biswas C: Stimulation of metalloproteinase production by recombinant EMMPRIN from transfected CHO cells. J Biol Chem 1996, 272:24-27 - PubMed
-
- Guo HL, Zucker S, Toole BP: EMMPRIN (CD147) an inducer of matrix metalloproteinase synthesis, also binds interstitial collagenase to the tumor cell surface. Cancer Res 2000, 60:888-891 - PubMed
-
- Basset P, Bellocq JP, Wolf C, Stoll I, Hutin P, Limacher JM, Podhajcer OL, Chenard MP, Rio MC, Chambon P: A novel metalloproteinase gene specifically expressed in stromal cells of breast carcinomas. Nature 1990, 348:699-704 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

