Internal aluminum block of plant inward K(+) channels
- PMID: 11402172
- PMCID: PMC135577
- DOI: 10.1105/tpc.13.6.1453
Internal aluminum block of plant inward K(+) channels
Abstract
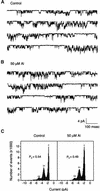
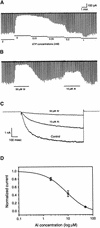
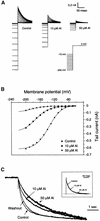
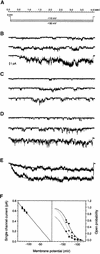
Aluminum (Al) inhibits inward K(+) channels (K(in)) in both root hair and guard cells, which accounts for at least part of the Al toxicity in plants. To understand the mechanism of Al-induced K(in) inhibition, we performed patch clamp analyses on K(in) in guard cells and on KAT1 channels expressed in Xenopus oocytes. Our results show that Al inhibits plant K(in) by blocking the channels at the cytoplasmic side of the plasma membrane. In guard cells, single-channel recording revealed that Al inhibition of K(in) occurred only upon internal exposure. Using both "giant patch" recording and single-channel analyses, we found that Al reduced KAT1 open probability and changed its activation kinetics through an internal membrane-delimited mechanism. We also provide evidence that a Ca(2)+ channel-like pathway that is sensitive to antagonists verapamil and La(3)+ mediates Al entry across the plasma membrane. We conclude that Al enters plant cells through a Ca(2)+ channel-like pathway and inhibits K(+) uptake by internally blocking K(in).
Figures





Similar articles
-
Distinct abscisic acid signaling pathways for modulation of guard cell versus mesophyll cell potassium channels revealed by expression studies in Xenopus laevis oocytes.Plant Physiol. 2000 Sep;124(1):223-30. doi: 10.1104/pp.124.1.223. Plant Physiol. 2000. PMID: 10982437 Free PMC article.
-
Co-expression of calcium-dependent protein kinase with the inward rectified guard cell K+ channel KAT1 alters current parameters in Xenopus laevis oocytes.Plant Cell Physiol. 2000 Jun;41(6):785-90. doi: 10.1093/pcp/41.6.785. Plant Cell Physiol. 2000. PMID: 10945349
-
Inward potassium channel in guard cells as a target for polyamine regulation of stomatal movements.Plant Physiol. 2000 Nov;124(3):1315-26. doi: 10.1104/pp.124.3.1315. Plant Physiol. 2000. PMID: 11080307 Free PMC article.
-
Inward rectifier potassium channels in plants differ from their animal counterparts in response to voltage and channel modulators.Eur Biophys J. 1995;24(2):107-15. doi: 10.1007/BF00211406. Eur Biophys J. 1995. PMID: 8582318
-
Evaluation of functional interaction between K(+) channel alpha- and beta-subunits and putative inactivation gating by Co-expression in Xenopus laevis oocytes.Plant Physiol. 1999 Nov;121(3):995-1002. doi: 10.1104/pp.121.3.995. Plant Physiol. 1999. PMID: 10557249 Free PMC article.
Cited by
-
Expressed sequence tag-based gene expression analysis under aluminum stress in rye.Plant Physiol. 2002 Dec;130(4):1706-16. doi: 10.1104/pp.009969. Plant Physiol. 2002. PMID: 12481053 Free PMC article.
-
A Ca(2)+ signaling pathway regulates a K(+) channel for low-K response in Arabidopsis.Proc Natl Acad Sci U S A. 2006 Aug 15;103(33):12625-30. doi: 10.1073/pnas.0605129103. Epub 2006 Aug 8. Proc Natl Acad Sci U S A. 2006. PMID: 16895985 Free PMC article.
-
Single mutations convert an outward K+ channel into an inward K+ channel.Proc Natl Acad Sci U S A. 2008 Feb 26;105(8):2871-6. doi: 10.1073/pnas.0712349105. Epub 2008 Feb 19. Proc Natl Acad Sci U S A. 2008. PMID: 18287042 Free PMC article.
-
Ion Flux in Roots of Chinese Fir (Cunninghamia lanceolata (Lamb.) Hook) under Aluminum Stress.PLoS One. 2016 Jun 6;11(6):e0156832. doi: 10.1371/journal.pone.0156832. eCollection 2016. PLoS One. 2016. PMID: 27270726 Free PMC article.
-
Potential of calcium nitrate to mitigate the aluminum toxicity in Phaseolus vulgaris: effects on morphoanatomical traits, mineral nutrition and photosynthesis.Ecotoxicology. 2020 Mar;29(2):203-216. doi: 10.1007/s10646-020-02168-6. Epub 2020 Jan 31. Ecotoxicology. 2020. PMID: 32006192
References
-
- Archambault, D.J., Zhang, G., and Taylor, G.J. (1997). Spatial variation in the kinetics of aluminium (Al) uptake in roots of wheat (Triticum aestivum L.) exhibiting differential resistance to Al: Evidence for metabolism-dependent exclusion of Al. J. Plant Physiol. 151, 668–674.
-
- Assmann, S.M. (1993). Signal transduction in guard cells. Annu. Rev. Cell Biol. 9, 345–375. - PubMed
-
- Bei, Q., and Luan, S. (1998). Functional expression and characterization of a plant K+ channel gene in a plant cell model. Plant J. 13, 857–865. - PubMed
-
- Boyer, J.S. (1985). Water transport. Annu. Rev. Plant Physiol. 36, 473–516.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

