Mice lacking tPA, uPA, or plasminogen genes showed delayed functional recovery after sciatic nerve crush
- PMID: 11404420
- PMCID: PMC6762762
- DOI: 10.1523/JNEUROSCI.21-12-04348.2001
Mice lacking tPA, uPA, or plasminogen genes showed delayed functional recovery after sciatic nerve crush
Abstract
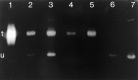
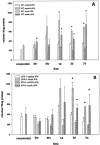
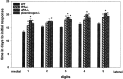
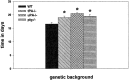
Axonal outgrowth during peripheral nerve regeneration relies on the ability of growth cones to traverse through an environment that has been altered structurally and along a basal lamina sheath to reinnervate synaptic targets. To promote migration, growth cones secrete proteases that are thought to dissolve cell-cell and cell-matrix adhesions. These proteases include the plasminogen activators (PAs), tissue PA (tPA) and urokinase PA (uPA), and their substrate, plasminogen. PA expression and secretion are upregulated in regenerating mammalian sensory neurons in culture. After sciatic nerve crush in mice, there was an induction of PA mRNAs in the sensory neurons contributing to the crushed nerve and an upregulation of PA-dependent activity in crushed nerve compared with sham counterparts during nerve regeneration. To further assess the role of the PA system during peripheral nerve regeneration, PA-dependent activity as well as recovery of sensory and motor function in the injured hindlimb were assessed in wild-type, tPA, uPA, and plasminogen knock-out mice. Protease activity visualized by gel zymography showed that after nerve crush, the upregulation of PA activity in the tPA and uPA knock-out mice was delayed compared with wild-type mice. Recovery of sensory function was assessed by toe pinch, footpad prick, and the toe-spreading reflex. All knock-out mice demonstrated a significant delay in hindlimb response to these sensory stimuli compared with wild-type mice. For each modality tested, the uPA knock-out mice were the most dramatically affected, showing the longest delay to initiate a response. These studies clearly showed that PAs were necessary for timely functional recovery by regenerating peripheral nerves.
Figures



Similar articles
-
Induction of the plasminogen activator system accompanies peripheral nerve regeneration after sciatic nerve crush.J Neurosci. 2001 Jun 15;21(12):4336-47. doi: 10.1523/JNEUROSCI.21-12-04336.2001. J Neurosci. 2001. PMID: 11404419 Free PMC article.
-
Mice lacking tissue plasminogen activator and urokinase plasminogen activator genes show attenuated matrix metalloproteases activity after sciatic nerve crush.J Neurosci Res. 2003 Nov 1;74(3):430-4. doi: 10.1002/jnr.10786. J Neurosci Res. 2003. PMID: 14598319
-
Mice without uPA, tPA, or plasminogen genes are resistant to experimental choroidal neovascularization.Invest Ophthalmol Vis Sci. 2003 Apr;44(4):1732-9. doi: 10.1167/iovs.02-0809. Invest Ophthalmol Vis Sci. 2003. PMID: 12657615
-
Induction of plasminogen activator inhibitor-1 and -2 in dorsal root ganglion neurons after peripheral nerve injury.Neuroscience. 2005;132(1):183-91. doi: 10.1016/j.neuroscience.2004.12.003. Neuroscience. 2005. PMID: 15780477
-
Tissue plasminogen activator in primary afferents induces dorsal horn excitability and pain response after peripheral nerve injury.Eur J Neurosci. 2004 Jan;19(1):93-102. doi: 10.1046/j.1460-9568.2003.03080.x. Eur J Neurosci. 2004. PMID: 14750967
Cited by
-
SH2B1beta (SH2-Bbeta) enhances expression of a subset of nerve growth factor-regulated genes important for neuronal differentiation including genes encoding urokinase plasminogen activator receptor and matrix metalloproteinase 3/10.Mol Endocrinol. 2008 Feb;22(2):454-76. doi: 10.1210/me.2007-0384. Epub 2007 Oct 18. Mol Endocrinol. 2008. PMID: 17947375 Free PMC article.
-
Induction of the plasminogen activator system accompanies peripheral nerve regeneration after sciatic nerve crush.J Neurosci. 2001 Jun 15;21(12):4336-47. doi: 10.1523/JNEUROSCI.21-12-04336.2001. J Neurosci. 2001. PMID: 11404419 Free PMC article.
-
tPA regulates neurite outgrowth by phosphorylation of LRP5/6 in neural progenitor cells.Mol Neurobiol. 2014 Feb;49(1):199-215. doi: 10.1007/s12035-013-8511-x. Epub 2013 Aug 8. Mol Neurobiol. 2014. PMID: 23925701
-
Bex1 is involved in the regeneration of axons after injury.J Neurochem. 2010 Nov;115(4):910-20. doi: 10.1111/j.1471-4159.2010.06960.x. Epub 2010 Sep 28. J Neurochem. 2010. PMID: 20731761 Free PMC article.
-
Plasminogen activator promotes recovery following spinal cord injury.Cell Mol Neurobiol. 2011 Aug;31(6):961-7. doi: 10.1007/s10571-011-9701-6. Epub 2011 May 14. Cell Mol Neurobiol. 2011. PMID: 21573723 Free PMC article.
References
-
- Avellino A, Hart D, Dailey A, Mackinnon M, Ellegala D, Kliot M. Differential macrophage responses in the peripheral and central nervous system during Wallerian degeneration of axons. Exp Neurol. 1995;136:183–198. - PubMed
-
- Azzouz M, Kennel P, Warter J, Poindron P, Borg J. Enhancement of mouse sciatic nerve regeneration by the long chain fatty alcohol, N-Hexacosanol. Exp Neurol. 1996;138:189–197. - PubMed
-
- Besser D, Verde P, Nagamine Y, Blasi F. Signal transduction and the uPA/u-PAR system. Fibrinolysis. 1996;10:215–237.
-
- Brauer P, Yee J. Cranial neural crest cells synthesize and secrete a latent form of transforming growth factor beta that can be activated by neural crest cell proteolysis. Dev Biol. 1993;155:281–285. - PubMed
-
- Carmeliet P, Collen D. Targeted gene manipulation and transfer of the plasminogen and coagulation systems in mice. Fibrinolysis. 1996;10:195–213. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
