Neutralizing monoclonal antibodies to hepatocyte growth factor/scatter factor (HGF/SF) display antitumor activity in animal models
- PMID: 11416216
- PMCID: PMC34688
- DOI: 10.1073/pnas.131200498
Neutralizing monoclonal antibodies to hepatocyte growth factor/scatter factor (HGF/SF) display antitumor activity in animal models
Abstract
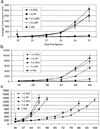
The hepatocyte growth factor (HGF/SF) receptor, Met, regulates mitogenesis, motility, and morphogenesis in a cell type-dependent fashion. Activation of Met via autocrine, paracrine, or mutational mechanisms can lead to tumorigenesis and metastasis and numerous studies have linked inappropriate expression of this ligand-receptor pair to most types of human solid tumors. To prepare mAbs to human HGF/SF, mice were immunized with native and denatured preparations of the ligand. Recloned mAbs were tested in vitro for blocking activity against scattering and branching morphogenesis. Our results show that no single mAb was capable of neutralizing the in vitro activity of HGF/SF, and that the ligand possesses a minimum of three epitopes that must be blocked to prevent Met tyrosine kinase activation. In vivo, the neutralizing mAb combination inhibited s.c. growth in athymic nu/nu mice of tumors dependent on an autocrine Met-HGF/SF loop. Importantly, growth of human glioblastoma multiforme xenografts expressing Met and HGF/SF were markedly reduced in the presence of HGF/SF-neutralizing mAbs. These results suggest interrupting autocrine and/or paracrine Met-HGF/SF signaling in tumors dependent on this pathway is a possible intervention strategy.
Figures

References
-
- Bottaro D P, Rubin J S, Faletto D L, Chan A M, Kmiecik T E, Vande Woude G F, Aaronson S A. Science. 1991;251:802–804. - PubMed
-
- Rubin J S, Bottaro D P, Aaronson S A. Biochim Biophys Acta. 1993;1155:357–371. - PubMed
-
- Tsarfaty I, Rong S, Resau J H, Shen R, Pedro Pinto D S, Vande Woude G F. Science. 1994;263:98–101. - PubMed
-
- Montesano R, Matsumoto K, Nakamura T, Orci L. Cell. 1991;67:901–908. - PubMed
-
- Tsarfaty I, Resau J H, Rulong S, Keydar I, Faletto D L, Vande Woude G F. Science. 1992;257:1258–1261. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

