Molecular basis of Ca(2)+ activation of the mouse cardiac Ca(2)+ release channel (ryanodine receptor)
- PMID: 11429443
- PMCID: PMC2233748
- DOI: 10.1085/jgp.118.1.33
Molecular basis of Ca(2)+ activation of the mouse cardiac Ca(2)+ release channel (ryanodine receptor)
Abstract
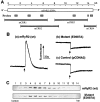
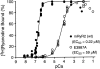
Activation of the cardiac ryanodine receptor (RyR2) by Ca(2)+ is an essential step in excitation-contraction coupling in heart muscle. However, little is known about the molecular basis of activation of RyR2 by Ca(2)+. In this study, we investigated the role in Ca(2)+ sensing of the conserved glutamate 3987 located in the predicted transmembrane segment M2 of the mouse RyR2. Single point mutation of this conserved glutamate to alanine (E3987A) reduced markedly the sensitivity of the channel to activation by Ca(2)+, as measured by using single-channel recordings in planar lipid bilayers and by [(3)H]ryanodine binding assay. However, this mutation did not alter the affinity of [(3)H]ryanodine binding and the single-channel conductance. In addition, the E3987A mutant channel was activated by caffeine and ATP, was inhibited by Mg(2)+, and was modified by ryanodine in a fashion similar to that of the wild-type channel. Coexpression of the wild-type and mutant E3987A RyR2 proteins in HEK293 cells produced individual single channels with intermediate sensitivities to activating Ca(2)+. These results are consistent with the view that glutamate 3987 is a major determinant of Ca(2)+ sensitivity to activation of the mouse RyR2 channel, and that Ca(2)+ sensing by RyR2 involves the cooperative action between ryanodine receptor monomers. The results of this study also provide initial insights into the structural and functional properties of the mouse RyR2, which should be useful for studying RyR2 function and regulation in genetically modified mouse models.
Figures






References
-
- Chamberlain B.K., Levitsky D.O., Fleischer S. Isolation and characterization of canine cardiac sarcoplasmic reticulum with improved Ca2+ transport properties. J. Biol. Chem. 1983;258:6602–6609. - PubMed
-
- Chen S.R., Ebisawa K., Li X., Zhang L. Molecular identification of the ryanodine receptor Ca2+ sensor. J. Biol. Chem. 1998;273:14675–14678. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

