Crystal structure of a superantigen bound to MHC class II displays zinc and peptide dependence
- PMID: 11432818
- PMCID: PMC125526
- DOI: 10.1093/emboj/20.13.3306
Crystal structure of a superantigen bound to MHC class II displays zinc and peptide dependence
Abstract
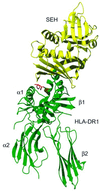
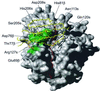
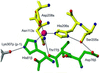
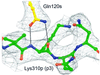
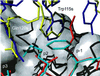
The three-dimensional structure of a bacterial superantigen, Staphylococcus aureus enterotoxin H (SEH), bound to human major histocompatibility complex (MHC) class II (HLA-DR1) has been determined by X-ray crystallography to 2.6 A resolution (1HXY). The superantigen binds on top of HLA-DR1 in a completely different way from earlier co-crystallized superantigens from S.aureus. SEH interacts with high affinity through a zinc ion with the beta1 chain of HLA-DR1 and also with the peptide presented by HLA-DR1. The structure suggests that all superantigens interacting with MHC class II in a zinc-dependent manner present the superantigen in a common way. This suggests a new model for ternary complex formation with the T-cell receptor (TCR), in which a contact between the TCR and the MHC class II is unlikely.
Figures

Similar articles
-
Three-dimensional structure of a human class II histocompatibility molecule complexed with superantigen.Nature. 1994 Apr 21;368(6473):711-8. doi: 10.1038/368711a0. Nature. 1994. PMID: 8152483
-
Crystal structure of staphylococcal enterotoxin I (SEI) in complex with a human major histocompatibility complex class II molecule.J Biol Chem. 2006 Sep 1;281(35):25356-64. doi: 10.1074/jbc.M603969200. Epub 2006 Jul 6. J Biol Chem. 2006. PMID: 16829512 Free PMC article.
-
The crystal structure of staphylococcal enterotoxin H: implications for binding properties to MHC class II and TcR molecules.J Mol Biol. 2000 Sep 22;302(3):527-37. doi: 10.1006/jmbi.2000.4093. J Mol Biol. 2000. PMID: 10986116
-
Superantigens. Gazing into the crystal ball.Curr Biol. 1995 Mar 1;5(3):235-7. doi: 10.1016/s0960-9822(95)00047-9. Curr Biol. 1995. PMID: 7780728 Review.
-
Superantigens: structure and relevance to human disease.Proc Soc Exp Biol Med. 1996 Jun;212(2):99-109. doi: 10.3181/00379727-212-43996. Proc Soc Exp Biol Med. 1996. PMID: 8650257 Review.
Cited by
-
egc Superantigens Impair Monocytes/Macrophages Inducing Cell Death and Inefficient Activation.Front Immunol. 2020 Jan 15;10:3008. doi: 10.3389/fimmu.2019.03008. eCollection 2019. Front Immunol. 2020. PMID: 32010128 Free PMC article.
-
Zinc induces dimerization of the class II major histocompatibility complex molecule that leads to cooperative binding to a superantigen.J Biol Chem. 2007 Mar 2;282(9):5991-6000. doi: 10.1074/jbc.M608482200. Epub 2006 Dec 13. J Biol Chem. 2007. PMID: 17166841 Free PMC article.
-
Microcalorimetric Investigations of Reversible Staphylococcal Enterotoxin Unfolding.Toxins (Basel). 2022 Aug 15;14(8):554. doi: 10.3390/toxins14080554. Toxins (Basel). 2022. PMID: 36006217 Free PMC article.
-
Staphylococcal enterotoxin H contrasts closely related enterotoxins in species reactivity.Immunology. 2002 May;106(1):71-9. doi: 10.1046/j.1365-2567.2002.01409.x. Immunology. 2002. PMID: 11972634 Free PMC article.
-
Control of established colon cancer xenografts using a novel humanized single chain antibody-streptococcal superantigen fusion protein targeting the 5T4 oncofetal antigen.PLoS One. 2014 Apr 15;9(4):e95200. doi: 10.1371/journal.pone.0095200. eCollection 2014. PLoS One. 2014. PMID: 24736661 Free PMC article.
References
-
- Al-Daccak R. et al. (1998) Staphylococcal enterotoxin D is a promiscuous superantigen offering multiple modes of interactions with the MHC class II receptors. J. Immunol., 160, 225–232. - PubMed
-
- Arcus V.L., Proft,T., Sigrill,J.A., Baker,H.M., Fraser,J.D. and Baker,E.N. (2000) Conservation and variation in superantigen structure and activity highlighted by the three-dimensional structure of two new superantigens from Streptococcus pyogenes. J. Mol. Biol., 299, 157–168. - PubMed
-
- Brünger A.T. et al. (1998) Crystallography & NMR system: a new software suite for macromolecular structure determination. Acta. Crystallogr. D, 54, 905–921. - PubMed
-
- Cerenius Y., Ståhl,K., Svensson,L.A., Ursby,T., Oskarsson,A., Albertsson,J. and Liljas,A. (2000) The crystallography beamline I711 at MAX II. J. Synchotron Radiat., 7, 203–208. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

