Double-check probing of DNA bending and unwinding by XPA-RPA: an architectural function in DNA repair
- PMID: 11432842
- PMCID: PMC125508
- DOI: 10.1093/emboj/20.13.3554
Double-check probing of DNA bending and unwinding by XPA-RPA: an architectural function in DNA repair
Abstract
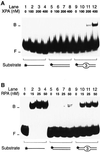
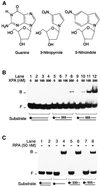
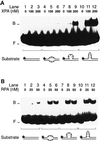
The multiprotein factor composed of XPA and replication protein A (RPA) is an essential subunit of the mammalian nucleotide excision repair system. Although XPA-RPA has been implicated in damage recognition, its activity in the DNA repair pathway remains controversial. By replacing DNA adducts with mispaired bases or non-hybridizing analogues, we found that the weak preference of XPA and RPA for damaged substrates is entirely mediated by indirect readout of DNA helix conformations. Further screening with artificially distorted substrates revealed that XPA binds most efficiently to rigidly bent duplexes but not to single-stranded DNA. Conversely, RPA recognizes single-stranded sites but not backbone bending. Thus, the association of XPA with RPA generates a double-check sensor that detects, simultaneously, backbone and base pair distortion of DNA. The affinity of XPA for sharply bent duplexes, characteristic of architectural proteins, is not compatible with a direct function during recognition of nucleotide lesions. Instead, XPA in conjunction with RPA may constitute a regulatory factor that monitors DNA bending and unwinding to verify the damage-specific localization of repair complexes or control their correct three-dimensional assembly.
Figures





Similar articles
-
Thermodynamic properties of damaged DNA and its recognition by xeroderma pigmentosum group A protein and replication protein A.Arch Biochem Biophys. 2006 Feb 1;446(1):1-10. doi: 10.1016/j.abb.2005.12.003. Epub 2005 Dec 22. Arch Biochem Biophys. 2006. PMID: 16405861
-
RPA involvement in the damage-recognition and incision steps of nucleotide excision repair.Nature. 1995 Apr 6;374(6522):566-9. doi: 10.1038/374566a0. Nature. 1995. PMID: 7700386
-
Recognition of nonhybridizing base pairs during nucleotide excision repair of DNA.Proc Natl Acad Sci U S A. 1999 May 25;96(11):6090-5. doi: 10.1073/pnas.96.11.6090. Proc Natl Acad Sci U S A. 1999. PMID: 10339546 Free PMC article.
-
Three-dimensional structural views of damaged-DNA recognition: T4 endonuclease V, E. coli Vsr protein, and human nucleotide excision repair factor XPA.Mutat Res. 2000 Aug 30;460(3-4):257-75. doi: 10.1016/s0921-8777(00)00031-8. Mutat Res. 2000. PMID: 10946233 Review.
-
Critical DNA damage recognition functions of XPC-hHR23B and XPA-RPA in nucleotide excision repair.Mol Carcinog. 2003 Sep;38(1):1-13. doi: 10.1002/mc.10143. Mol Carcinog. 2003. PMID: 12949838 Review.
Cited by
-
Emerging Roles of Post-Translational Modifications in Nucleotide Excision Repair.Cells. 2020 Jun 15;9(6):1466. doi: 10.3390/cells9061466. Cells. 2020. PMID: 32549338 Free PMC article. Review.
-
Cooperative interaction of human XPA stabilizes and enhances specific binding of XPA to DNA damage.Biochemistry. 2005 May 17;44(19):7361-8. doi: 10.1021/bi047598y. Biochemistry. 2005. PMID: 15882075 Free PMC article.
-
Dimerization of human XPA and formation of XPA2-RPA protein complex.Biochemistry. 2002 Oct 29;41(43):13012-20. doi: 10.1021/bi026064z. Biochemistry. 2002. PMID: 12390028 Free PMC article.
-
Xeroderma pigmentosum group A protein loads as a separate factor onto DNA lesions.Mol Cell Biol. 2003 Aug;23(16):5755-67. doi: 10.1128/MCB.23.16.5755-5767.2003. Mol Cell Biol. 2003. PMID: 12897146 Free PMC article.
-
Human HMGB1 directly facilitates interactions between nucleotide excision repair proteins on triplex-directed psoralen interstrand crosslinks.DNA Repair (Amst). 2009 Jul 4;8(7):865-72. doi: 10.1016/j.dnarep.2009.04.001. Epub 2009 May 14. DNA Repair (Amst). 2009. PMID: 19446504 Free PMC article.
References
-
- Aboussekhra A. et al. (1995) Mammalian DNA nucleotide excision repair reconstituted with purified components. Cell, 80, 859–868. - PubMed
-
- Araújo S.J., Tirode,F., Coin,F., Pospiech,H., Syväoja,J.E., Stucki,M., Hübscher,U., Egly,J.-M. and Wood,R.D. (2000) Nucleotide excision repair of DNA with recombinant human proteins: definition of the minimal set of factors, active forms of TFIIH, and modulation by CAK. Genes Dev., 14, 349–359. - PMC - PubMed
-
- Asahina H., Kuraoka,I., Shirakawa,M., Morita,E.H., Miura,N., Miyamoto,I., Ohtsuka,E., Okada,Y. and Tanaka,K. (1994) The XPA protein is a zinc metalloprotein with an ability to recognize various kinds of DNA damage. Mutat. Res., 315, 229–237. - PubMed
-
- Batty D., Rapic’-Otrin,V., Levine,A.S. and Wood,R.D. (2000) Stable binding of human XPC complex to irradiated DNA confers strong discrimination for damaged sites. J. Mol. Biol., 300, 275–290. - PubMed
-
- Burns J.L., Guzder,S.N., Sung,P., Prakash,S. and Prakash,L. (1996) An affinity of human replication protein A for ultraviolet-damaged DNA. J. Biol. Chem., 271, 11607–11610. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

