Porphyromonas gingivalis fimbriae inhibit caspase-3-mediated apoptosis of monocytic THP-1 cells under growth factor deprivation via extracellular signal-regulated kinase-dependent expression of p21 Cip/WAF1
- PMID: 11447172
- PMCID: PMC98586
- DOI: 10.1128/IAI.69.8.4944-4950.2001
Porphyromonas gingivalis fimbriae inhibit caspase-3-mediated apoptosis of monocytic THP-1 cells under growth factor deprivation via extracellular signal-regulated kinase-dependent expression of p21 Cip/WAF1
Abstract
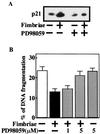
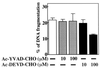
Apoptotic regulation of monocytes/macrophages appears to be closely associated with chronic inflammatory reactions. Since it was demonstrated earlier that certain bacterial cell components are involved in apoptotic regulation of these cells, in the present study, we investigated whether the bacterial fimbria, an important cell structure involved in bacterial adherence to host cells, regulates apoptosis of human monocytic THP-1 cells induced under growth factor deprivation. To investigate this point, we used fimbriae of Porphyromonas gingivalis, a pathogen causing periodontal disease, which is a chronic inflammatory disease. The fimbriae inhibited apoptosis of the cells under growth factor deprivation. This inhibitory action of the fimbriae was completely neutralized by anti-fimbrial antibody. The fimbriae stimulated activation of extracellular signal-regulated kinase (ERK) and expression of cyclin-dependent kinase inhibitor p21 Cip/WAF1 (p21) in the cells. The stimulatory effect of the fimbriae on the expression of the p21 protein was inhibited by treatment with PD98059, a specific inhibitor of ERK. The cell apoptosis was inhibited by treatment with Ac-DEVD-CHO, an inhibitor of caspase-3. The fimbriae inhibited the serum withdrawal-induced cleavage of the caspase-3 proform and poly(ADP-ribose) polymerase, one of the caspase-3 substrates. Furthermore, PD98059 and antisense p21 oligonucleotide blocked the fimbrial inhibition of apoptosis and caspase-3 activation of the cells induced by serum withdrawal. These results show that the bacterial fimbriae inhibited apoptosis of THP-1 cells induced under growth factor deprivation via ERK-dependent expression of p21. The present study suggests that bacterial fimbriae act as potent regulators of chronic inflammatory disease, e.g., periodontal disease, through blocking apoptosis of monocytes/macrophages.
Figures






Similar articles
-
Clostridium difficile toxin A-induced colonocyte apoptosis involves p53-dependent p21(WAF1/CIP1) induction via p38 mitogen-activated protein kinase.Gastroenterology. 2005 Dec;129(6):1875-88. doi: 10.1053/j.gastro.2005.09.011. Gastroenterology. 2005. PMID: 16344056
-
Activation of extracellular signal-regulated kinases ERK1 and ERK2 induces Bcl-xL up-regulation via inhibition of caspase activities in erythropoietin signaling.J Cell Physiol. 2003 May;195(2):290-7. doi: 10.1002/jcp.10245. J Cell Physiol. 2003. PMID: 12652655
-
Indomethacin induces apoptosis in 786-O renal cell carcinoma cells by activating mitogen-activated protein kinases and AKT.Eur J Pharmacol. 2007 Jun 1;563(1-3):49-60. doi: 10.1016/j.ejphar.2007.01.071. Epub 2007 Feb 8. Eur J Pharmacol. 2007. PMID: 17341418
-
Caspase-mediated activation of PAK2 during apoptosis: proteolytic kinase activation as a general mechanism of apoptotic signal transduction?Cell Death Differ. 1998 Aug;5(8):637-45. doi: 10.1038/sj.cdd.4400405. Cell Death Differ. 1998. PMID: 10200518 Review.
-
Fimbriae, transmembrane signaling, and cell activation.J Infect Dis. 2001 Mar 1;183 Suppl 1:S47-50. doi: 10.1086/318851. J Infect Dis. 2001. PMID: 11171014 Review. No abstract available.
Cited by
-
Apoptosis and its pathways as targets for intracellular pathogens to persist in cells.Parasitol Res. 2023 Dec 19;123(1):60. doi: 10.1007/s00436-023-08031-x. Parasitol Res. 2023. PMID: 38112844 Free PMC article. Review.
-
Activation of the phosphatidylinositol 3-kinase/Akt pathway contributes to survival of primary epithelial cells infected with the periodontal pathogen Porphyromonas gingivalis.Infect Immun. 2004 Jul;72(7):3743-51. doi: 10.1128/IAI.72.7.3743-3751.2004. Infect Immun. 2004. PMID: 15213114 Free PMC article.
-
Purification and characterization of a fimbrial protein from Porphyromonas salivosa ATCC 49407.J Vet Med Sci. 2019 Jun 28;81(6):916-923. doi: 10.1292/jvms.19-0067. Epub 2019 Apr 23. J Vet Med Sci. 2019. PMID: 31019151 Free PMC article.
-
Gingipain-dependent interactions with the host are important for survival of Porphyromonas gingivalis.Front Biosci. 2008 May 1;13:3215-38. doi: 10.2741/2922. Front Biosci. 2008. PMID: 18508429 Free PMC article. Review.
-
The chronicles of Porphyromonas gingivalis: the microbium, the human oral epithelium and their interplay.Microbiology (Reading). 2008 Oct;154(Pt 10):2897-2903. doi: 10.1099/mic.0.2008/021220-0. Microbiology (Reading). 2008. PMID: 18832296 Free PMC article. Review.
References
-
- Albina J E, Cui S, Mateo R B, Reichner J S. Nitric oxide-mediated apoptosis in murine peritoneal macrophages. J Immunol. 1993;150:5080–5085. - PubMed
-
- Asada M, Yamada T, Fukumuro K, Mizutani S. p21Cip/WAF1 is important for differentiation and survival of U937 cells. Leukemia. 1998;12:1944–1950. - PubMed
-
- Ashkenazi A, Dixit V M. Death receptors: signaling and modulation. Science. 1998;281:1305–1308. - PubMed
-
- Auer K L, Spector M S, Tombes R M, Seth P, Fisher P B, Gao B, Dent P, Kunos G. α-Adrenergic inhibiton of proliferation in HepG2 cells stably transfected with the α1B-adrenergic receptor through a p42MAP kinase/p21Cip1/WAF1-dependent pathway. FEBS Lett. 1998;436:131–138. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

