Arginine-specific protease from Porphyromonas gingivalis activates protease-activated receptors on human oral epithelial cells and induces interleukin-6 secretion
- PMID: 11447194
- PMCID: PMC98608
- DOI: 10.1128/IAI.69.8.5121-5130.2001
Arginine-specific protease from Porphyromonas gingivalis activates protease-activated receptors on human oral epithelial cells and induces interleukin-6 secretion
Abstract
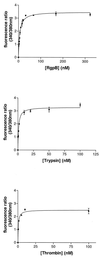
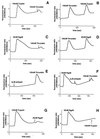
Periodontitis is a chronic inflammatory disease affecting oral tissues. Oral epithelial cells represent the primary barrier against bacteria causing the disease. We examined the responses of such cells to an arginine-specific cysteine proteinase (RgpB) produced by a causative agent of periodontal disease, Porphyromonas gingivalis. This protease caused an intracellular calcium transient in an oral epithelial cell line (KB), which was dependent on its enzymatic activity. Since protease-activated receptors (PARs) might mediate such signaling, reverse transcription-PCR was used to characterize the range of these receptors expressed in the KB cells. The cells were found to express PAR-1, PAR-2, and PAR-3, but not PAR-4. In immunohistochemical studies, human gingival epithelial cells were found to express PAR-1, PAR-2, and PAR-3 on their surface, but not PAR-4, indicating that the cell line was an effective model for the in vivo situation. PAR-1 and PAR-2 expression was confirmed in intracellular calcium mobilization assays by treatment of the cells with the relevant receptor agonist peptides. Desensitization experiments strongly indicated that signaling of the effects of RgpB was occurring through PAR-1 and PAR-2. Studies with cells individually transfected with each of these two receptors confirmed that they were both activated by RgpB. Finally, it was shown that, in the oral epithelial cell line, PAR activation by the bacterial protease-stimulated secretion of interleukin-6. This induction of a powerful proinflammatory cytokine suggests a mechanism whereby cysteine proteases from P. gingivalis might mediate inflammatory events associated with periodontal disease on first contact with a primary barrier of cells.
Figures





Similar articles
-
Activation of protease-activated receptors by gingipains from Porphyromonas gingivalis leads to platelet aggregation: a new trait in microbial pathogenicity.Blood. 2001 Jun 15;97(12):3790-7. doi: 10.1182/blood.v97.12.3790. Blood. 2001. PMID: 11389018
-
Protease-activated receptor signaling increases epithelial antimicrobial peptide expression.J Immunol. 2004 Oct 15;173(8):5165-70. doi: 10.4049/jimmunol.173.8.5165. J Immunol. 2004. PMID: 15470061
-
Cleavage of protease-activated receptors on an immortalized oral epithelial cell line by Porphyromonas gingivalis gingipains.Microbiology (Reading). 2009 Oct;155(Pt 10):3238-3246. doi: 10.1099/mic.0.029132-0. Epub 2009 Jul 16. Microbiology (Reading). 2009. PMID: 19608609 Free PMC article.
-
The Role of Proteinase-Activated Receptors 1 and 2 in the Regulation of Periodontal Tissue Metabolism and Disease.J Immunol Res. 2017;2017:5193572. doi: 10.1155/2017/5193572. Epub 2017 Apr 19. J Immunol Res. 2017. PMID: 28503577 Free PMC article. Review.
-
The role of gingipains in the pathogenesis of periodontal disease.J Periodontol. 2003 Jan;74(1):111-8. doi: 10.1902/jop.2003.74.1.111. J Periodontol. 2003. PMID: 12593605 Review.
Cited by
-
Periodontal bacterial supernatants modify differentiation, migration and inflammatory cytokine expression in human periodontal ligament stem cells.PLoS One. 2019 Jul 3;14(7):e0219181. doi: 10.1371/journal.pone.0219181. eCollection 2019. PLoS One. 2019. PMID: 31269072 Free PMC article.
-
Periodontitis: Grade Modifiers Revisited.Oral Dis. 2025 Jun;31(6):1637-1646. doi: 10.1111/odi.15297. Epub 2025 Feb 27. Oral Dis. 2025. PMID: 40013676 Free PMC article. Review.
-
Serine Protease-Mediated Cutaneous Inflammation: Characterization of an Ex Vivo Skin Model for the Assessment of Dexamethasone-Loaded Core Multishell-Nanocarriers.Pharmaceutics. 2020 Sep 10;12(9):862. doi: 10.3390/pharmaceutics12090862. Pharmaceutics. 2020. PMID: 32927792 Free PMC article.
-
Distribution and factors associated with salivary secretory leukocyte protease inhibitor concentrations.Oral Dis. 2016 Nov;22(8):781-790. doi: 10.1111/odi.12550. Epub 2016 Aug 17. Oral Dis. 2016. PMID: 27470907 Free PMC article.
-
Bacterial invasion of epithelial cells and spreading in periodontal tissue.Periodontol 2000. 2010 Feb;52(1):68-83. doi: 10.1111/j.1600-0757.2009.00323.x. Periodontol 2000. 2010. PMID: 20017796 Free PMC article. Review. No abstract available.
References
-
- Abraham L A, Jenkins A L, Stone S R, Mackie E J. Expression of the thrombin receptor in developing bone and associated tissues. J Bone Miner Res. 1998;13:818–827. - PubMed
-
- Andrade-Gordon P, Maryanoff B E, Derian C K, Zhang H C, Addo M F, Darrow A L, Eckardt A J, Hoekstra W J, McComsey D F, Oksenberg D, Reynolds E E, Santulli R J, Scarborough R M, Smith C E, White K B. Design, synthesis, and biological characterization of a peptide-mimetic antagonist for a tethered-ligand receptor. Proc Natl Acad Sci USA. 1999;96:12257–12262. - PMC - PubMed
-
- Bootman M D, Taylor C W, Berridge M J. The thiol reagent, thimerosal, evokes Ca2+ spikes in HeLa cells by sensitizing the inositol 1,4,5-trisphosphate receptor. J Biol Chem. 1992;267:25113–25119. - PubMed
-
- Chen C C, Chang K L, Huang J F, Huang J S, Tsai C C. Correlation of interleukin-1 beta, interleukin-6, and periodontitis. Kaohsiung J Med Sci. 1997;13:609–617. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

