RhoA is required for monocyte tail retraction during transendothelial migration
- PMID: 11448997
- PMCID: PMC2196864
- DOI: 10.1083/jcb.200103048
RhoA is required for monocyte tail retraction during transendothelial migration
Abstract
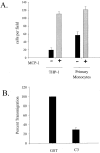
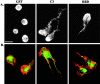
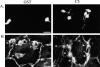
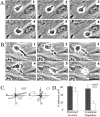
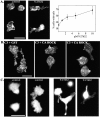
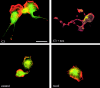
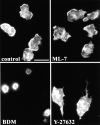
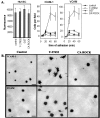
Transendothelial migration of monocytes is the process by which monocytes leave the circulatory system and extravasate through the endothelial lining of the blood vessel wall and enter the underlying tissue. Transmigration requires coordination of alterations in cell shape and adhesive properties that are mediated by cytoskeletal dynamics. We have analyzed the function of RhoA in the cytoskeletal reorganizations that occur during transmigration. By loading monocytes with C3, an inhibitor of RhoA, we found that RhoA was required for transendothelial migration. We then examined individual steps of transmigration to explore the requirement for RhoA in extravasation. Our studies showed that RhoA was not required for monocyte attachment to the endothelium nor subsequent spreading of the monocyte on the endothelial surface. Time-lapse video microscopy analysis revealed that C3-loaded monocytes also had significant forward crawling movement on the endothelial monolayer and were able to invade between neighboring endothelial cells. However, RhoA was required to retract the tail of the migrating monocyte and complete diapedesis. We also demonstrate that p160ROCK, a serine/threonine kinase effector of RhoA, is both necessary and sufficient for RhoA-mediated tail retraction. Finally, we find that p160ROCK signaling negatively regulates integrin adhesions and that inhibition of RhoA results in an accumulation of beta2 integrin in the unretracted tails.
Figures

Similar articles
-
RhoA activation promotes transendothelial migration of monocytes via ROCK.J Leukoc Biol. 2004 Mar;75(3):523-8. doi: 10.1189/jlb.0203054. Epub 2003 Nov 21. J Leukoc Biol. 2004. PMID: 14634067
-
Role of RhoA and Rho kinase in lysophosphatidic acid-induced endothelial barrier dysfunction.Arterioscler Thromb Vasc Biol. 2000 Dec;20(12):E127-33. doi: 10.1161/01.atv.20.12.e127. Arterioscler Thromb Vasc Biol. 2000. PMID: 11116077
-
Activation of RhoA by thrombin in endothelial hyperpermeability: role of Rho kinase and protein tyrosine kinases.Circ Res. 2000 Aug 18;87(4):335-40. doi: 10.1161/01.res.87.4.335. Circ Res. 2000. PMID: 10948069
-
Small guanine nucleotide-binding protein Rho and myocardial function.Acta Pharmacol Sin. 2005 Mar;26(3):279-85. doi: 10.1111/j.1745-7254.2005.00059.x. Acta Pharmacol Sin. 2005. PMID: 15715922 Review.
-
Leukocyte transendothelial migration: orchestrating the underlying molecular machinery.Curr Opin Cell Biol. 2001 Oct;13(5):569-77. doi: 10.1016/s0955-0674(00)00253-2. Curr Opin Cell Biol. 2001. PMID: 11544025 Review.
Cited by
-
Attenuation of monocyte chemotaxis--a novel anti-inflammatory mechanism of action for the cardio-protective hormone B-type natriuretic peptide.J Cardiovasc Transl Res. 2013 Aug;6(4):545-57. doi: 10.1007/s12265-013-9456-1. Epub 2013 Apr 27. J Cardiovasc Transl Res. 2013. PMID: 23625718
-
Type Igamma PIP kinase is a novel uropod component that regulates rear retraction during neutrophil chemotaxis.Mol Biol Cell. 2007 Dec;18(12):5069-80. doi: 10.1091/mbc.e07-05-0428. Epub 2007 Oct 10. Mol Biol Cell. 2007. PMID: 17928408 Free PMC article.
-
Plasma membrane organization is essential for balancing competing pseudopod- and uropod-promoting signals during neutrophil polarization and migration.Mol Biol Cell. 2005 Dec;16(12):5773-83. doi: 10.1091/mbc.e05-04-0358. Epub 2005 Oct 5. Mol Biol Cell. 2005. PMID: 16207810 Free PMC article.
-
PSGL-1 and mTOR regulate translation of ROCK-1 and physiological functions of macrophages.EMBO J. 2007 Jan 24;26(2):505-15. doi: 10.1038/sj.emboj.7601522. EMBO J. 2007. PMID: 17245434 Free PMC article.
-
A translocated effector required for Bartonella dissemination from derma to blood safeguards migratory host cells from damage by co-translocated effectors.PLoS Pathog. 2014 Jun 19;10(6):e1004187. doi: 10.1371/journal.ppat.1004187. eCollection 2014 Jun. PLoS Pathog. 2014. PMID: 24945914 Free PMC article.
References
-
- Aepfelbacher, M. 1995. ADP-ribosylation of Rho enhances adhesion of U937 cells to fibronectin via the α 5 β 1 integrin receptor. FEBS Lett. 363:78–80. - PubMed
-
- Aepfelbacher, M., M. Essler, E. Huber, A. Czech, and P.C. Weber. 1996. Rho is a negative regulator of human monocyte spreading. J. Immunol. 157:5070–5075. - PubMed
-
- Allen, W.E., G.E. Jones, J.W. Pollard, and A.J. Ridley. 1997. Rho, Rac and Cdc42 regulate actin organization and cell adhesion in macrophages. J. Cell Sci. 110:707–720. - PubMed
-
- Amano, M., Y. Fukata, and K. Kaibuchi. 2000. Regulation and functions of Rho-associated kinase. Exp Cell Res. 261:44–51. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

