Nitric oxide and bone
- PMID: 11454054
- PMCID: PMC1783253
- DOI: 10.1046/j.1365-2567.2001.01261.x
Nitric oxide and bone
Abstract
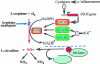
Nitric oxide (NO) is a free radical which has important effects on bone cell function. The endothelial isoform of nitric oxide synthase (eNOS) is widely expressed in bone on a constitutive basis, whereas inducible NOS is only expressed in response to inflammatory stimuli. It is currently unclear whether neuronal NOS is expressed by bone cells. Pro-inflammatory cytokines such as IL-1 and TNF cause activation of the iNOS pathway in bone cells and NO derived from this pathway potentiates cytokine and inflammation induced bone loss. These actions of NO are relevant to the pathogenesis of osteoporosis in inflammatory diseases such as rheumatoid arthritis, which are characterized by increased NO production and cytokine activation. Interferon gamma is a particularly potent stimulator of NO production when combined with other cytokines, causing very high concentrations of NO to be produced. These high levels of NO inhibit bone resorption and formation and may act to suppress bone turnover in severe inflammation. The eNOS isoform seems to play a key role in regulating osteoblast activity and bone formation since eNOS knockout mice have osteoporosis due to defective bone formation. Other studies have indicated that the NO derived from the eNOS pathway acts as a mediator of the effects of oestrogen in bone. eNOS also mediates the effects of mechanical loading on the skeleton where it acts along with prostaglandins, to promote bone formation and suppress bone resorption. Pharmacological NO donors have been shown to increase bone mass in experimental animals and preliminary evidence suggests that these agents may also influence bone turnover in man. These data indicate that the L-arginine/NO pathway represents a novel target for therapeutic intervention in the prevention and treatment of bone diseases.
Figures

Similar articles
-
The Michael Mason Prize Essay 1997. Nitric oxide and bone: what a gas!Br J Rheumatol. 1997 Aug;36(8):831-8. doi: 10.1093/rheumatology/36.8.831. Br J Rheumatol. 1997. PMID: 9291850 Review.
-
Nitric oxide and bone.J Bone Miner Res. 1996 Mar;11(3):300-5. doi: 10.1002/jbmr.5650110303. J Bone Miner Res. 1996. PMID: 8852940 Review.
-
A possible role for nitric oxide in osteoclastogenesis associated with cholesteatoma.Otol Neurotol. 2004 Sep;25(5):661-8. doi: 10.1097/00129492-200409000-00003. Otol Neurotol. 2004. PMID: 15353992
-
Defective bone formation and anabolic response to exogenous estrogen in mice with targeted disruption of endothelial nitric oxide synthase.Endocrinology. 2001 Feb;142(2):760-6. doi: 10.1210/endo.142.2.7977. Endocrinology. 2001. PMID: 11159848
-
Mechanical strain stimulates nitric oxide production by rapid activation of endothelial nitric oxide synthase in osteocytes.J Bone Miner Res. 1999 Jul;14(7):1123-31. doi: 10.1359/jbmr.1999.14.7.1123. J Bone Miner Res. 1999. PMID: 10404012
Cited by
-
Mineralized DNA-collagen complex-based biomaterials for bone tissue engineering.Int J Biol Macromol. 2020 Oct 15;161:1127-1139. doi: 10.1016/j.ijbiomac.2020.06.126. Epub 2020 Jun 16. Int J Biol Macromol. 2020. PMID: 32561285 Free PMC article.
-
Changes in Bone Marrow Fatty Acids Early after Ovariectomy-Induced Osteoporosis in Rats and Potential Functions.Metabolites. 2022 Dec 26;13(1):36. doi: 10.3390/metabo13010036. Metabolites. 2022. PMID: 36676961 Free PMC article.
-
Nitric oxide and inflammatory mediators in the perpetuation of osteoarthritis.Curr Rheumatol Rep. 2001 Dec;3(6):535-41. doi: 10.1007/s11926-001-0069-3. Curr Rheumatol Rep. 2001. PMID: 11709117 Review.
-
Nitric oxide and cyclic GMP functions in bone.Nitric Oxide. 2018 Jun 1;76:62-70. doi: 10.1016/j.niox.2018.03.007. Epub 2018 Mar 14. Nitric Oxide. 2018. PMID: 29550520 Free PMC article. Review.
-
miR-29b-3p regulated osteoblast differentiation via regulating IGF-1 secretion of mechanically stimulated osteocytes.Cell Mol Biol Lett. 2019 Mar 14;24:11. doi: 10.1186/s11658-019-0136-2. eCollection 2019. Cell Mol Biol Lett. 2019. PMID: 30915127 Free PMC article.
References
-
- Palmer RM, Ferrige AG, Moncada S. Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature. 1987;327:524–6. - PubMed
-
- Moncada S, Higgs A. The L-arginine nitric oxide pathway. N Engl J Med. 1993;329:2002–12. - PubMed
-
- Teitelbaum SL. Bone resorption by osteoclasts. Science. 2000;289:1504–8. 10.1126/science.289.5484.1504. - DOI - PubMed
-
- MacDonald BR, Takahashi N, McManus LM, et al. Formation of multinucleated cells that respond to osteotropic hormones in long-term human marrow cultures. Endocrinology. 1987;120:2326–33. - PubMed
-
- Tsukii K, Shima N, Mochizuki S, Yamaguchi K, Kinosaki M, Yano K, Shibata O, Udagawa N, Yasuda H, Suda T, Higashio K. Osteoclast differentiation factor mediates an essential signal for bone resorption induced by 1 alpha,25-dihydroxyvitamin D3, prostaglandin E2, or parathyroid hormone in the microenvironment of bone. Biochem Biophys Res Comm. 1998;246:337–41. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

