The role of donor T cells for target organ injuries in acute and chronic graft-versus-host disease
- PMID: 11454060
- PMCID: PMC1783249
- DOI: 10.1046/j.1365-2567.2001.01240.x
The role of donor T cells for target organ injuries in acute and chronic graft-versus-host disease
Abstract
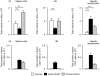
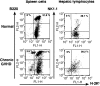
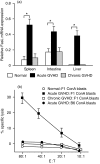
Donor T cells are crucial for target organ injury in graft-versus-host disease (GVHD). We examined the effects of donor T cells on the target organs using a parent-into-F1 model of acute and chronic GVHD. Donor T cells showed engraftment in the spleen, small intestine and liver of mice with acute GVHD, causing typical GVHD pathology in these organs. Interferon-gamma and Fas ligand expression were up-regulated, and host lymphocytes were depleted in the target organs of these mice. In contrast, donor T cells did not show engraftment in the small intestine of mice with chronic GVHD, and no GVHD pathology was observed in this organ. However, both donor T-cell engraftment and GVHD pathology were observed in the spleen and liver of chronic GVHD mice, along with the up-regulation of interleukin-4 (IL-4) and IL-10 expression plus the expansion of host lymphocytes such as splenic B cells and hepatic natural killer (NK) 1.1+ T cells. Donor anti-host cytotoxic T-lymphocyte activity was observed in spleen cells from mice with acute GVHD, but not in spleen cells from mice with chronic GVHD. Transplantation of Fas ligand-deficient (gld) spleen cells did not induce host lymphocyte depletion in target organs. These results indicate that donor T cells augment type 1 T helper immune responses and deplete the host lymphocytes from target organs mainly by Fas-mediated pathways in acute GVHD, while donor T cells augment type 2 T helper immune responses and expand host splenic B cells and hepatic NK1.1+ T cells in chronic GVHD.
Figures



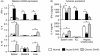
Similar articles
-
Differential expression of Fas and Fas ligand in acute and chronic graft-versus-host disease: up-regulation of Fas and Fas ligand requires CD8+ T cell activation and IFN-gamma production.J Immunol. 1998 Sep 15;161(6):2848-55. J Immunol. 1998. PMID: 9743345
-
Graft-versus-host-disease-associated donor cell engraftment in an F1 hybrid model is dependent upon the Fas pathway.Immunology. 2000 Jan;99(1):94-100. doi: 10.1046/j.1365-2567.2000.00919.x. Immunology. 2000. PMID: 10651946 Free PMC article.
-
Phenotypic analysis of donor cells infiltrating the small intestinal epithelium and spleen during graft-versus-host disease.Transplantation. 1995 Jan 27;59(2):268-73. Transplantation. 1995. PMID: 7839451
-
Th2 and Tc2 cells in the regulation of GVHD, GVL, and graft rejection: considerations for the allogeneic transplantation therapy of leukemia and lymphoma.Leuk Lymphoma. 2000 Jul;38(3-4):221-34. doi: 10.3109/10428190009087014. Leuk Lymphoma. 2000. PMID: 10830730 Review.
-
Graft-versus-host disease and the Th1/Th2 paradigm.Immunol Res. 1996;15(1):50-73. doi: 10.1007/BF02918284. Immunol Res. 1996. PMID: 8739565 Review.
Cited by
-
Mesenchymal stromal cells treatment attenuates dry eye in patients with chronic graft-versus-host disease.Mol Ther. 2012 Dec;20(12):2347-54. doi: 10.1038/mt.2012.208. Epub 2012 Oct 16. Mol Ther. 2012. PMID: 23070118 Free PMC article. Clinical Trial.
-
Inhibition of donor-derived T cells trafficking into target organs by FTY720 during acute graft-versus-host disease in small bowel transplantation.Clin Exp Immunol. 2006 Oct;146(1):85-92. doi: 10.1111/j.1365-2249.2006.03175.x. Clin Exp Immunol. 2006. PMID: 16968402 Free PMC article.
-
Expansion and activation kinetics of immune cells during early phase of GVHD in mouse model based on chemotherapy conditioning.Clin Dev Immunol. 2010;2010:142943. doi: 10.1155/2010/142943. Epub 2010 Dec 21. Clin Dev Immunol. 2010. PMID: 21197273 Free PMC article.
-
Biology of chronic graft-vs-host disease: Immune mechanisms and progress in biomarker discovery.World J Transplant. 2016 Dec 24;6(4):608-619. doi: 10.5500/wjt.v6.i4.608. World J Transplant. 2016. PMID: 28058210 Free PMC article. Review.
-
Contrasting acute graft-versus-host disease effects of Tim-3/galectin-9 pathway blockade dependent upon the presence of donor regulatory T cells.Blood. 2012 Jul 19;120(3):682-90. doi: 10.1182/blood-2011-10-387977. Epub 2012 Jun 7. Blood. 2012. PMID: 22677125 Free PMC article.
References
-
- Prentice HG, Blacklock HA, Janossy G, Bradstock KF, Skeggs D, Goldstein G, Hoffbrand AV. Use of anti-T cell monoclonal antibody OKT3 to prevent acute graft-versus-host disease in allogeneic bone marrow transplantation for acute leukemia. Lancet. 1982;1:310–318. - PubMed
-
- Blazer BR, Taylor PA, Snover DC, Bluestone JA, Vallera DA. Nonmitogenic anti-CD3F (ab′) 2 fragments inhibit lethal murine graft-versus-host disease induced across the major histocompatibility barrier. J Immunol. 1993;150:265–77. - PubMed
-
- Hakim FT, Mackall CL. The immune system: Effector and target of graft-versus-host disease. In: Ferrara JLM, Deeg HJ, Burakoff SJ, editors. Graft-Vs-Host Disease. 2. New York: Marcel Dekker, Inc.; 1997. pp. 257–89.
-
- Rozendaal L, Pals ST, Schilham M, Melief CJ, Gleichmann E. Allosuppression of B cells in vitro by graft-versus-host reaction-derived T cells is caused by cytotoxic T lymphocytes. Eur J Immunol. 1989;19:1669–75. - PubMed
-
- Rus V, Svetic A, Nguyen PH, Gause WC, Via CS. Kinetics of Th1 and Th2 cytokine production during the early course of acute and chronic murine graft-versus-host disease: Regulatory role of donor CD8+ T cells. J Immunol. 1995;155:2396–406. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

