Streptococcal inhibitor of complement (SIC) inhibits the membrane attack complex by preventing uptake of C567 onto cell membranes
- PMID: 11454069
- PMCID: PMC1783247
- DOI: 10.1046/j.1365-2567.2001.01249.x
Streptococcal inhibitor of complement (SIC) inhibits the membrane attack complex by preventing uptake of C567 onto cell membranes
Abstract
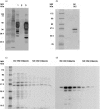
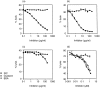
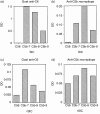
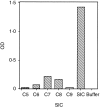
Streptococcal inhibitor of complement (SIC) was first described in 1996 as a putative inhibitor of the membrane attack complex of complement (MAC). SIC is a 31 000 MW protein secreted in large quantities by the virulent Streptococcus pyogenes strains M1 and M57, and is encoded by a gene which is extremely variable. In order to study further the interactions of SIC with the MAC, we have made a recombinant form of SIC (rSIC) in Escherichia coli and purified native M1 SIC which was used to raise a polyclonal antibody. SIC prevented reactive lysis of guinea pig erythrocytes by the MAC at a stage prior to C5b67 complexes binding to cell membranes, presumably by blocking the transiently expressed membrane insertion site on C7. The ability of SIC and clusterin (another putative fluid phase complement inhibitor) to inhibit complement lysis was compared, and found to be equally efficient. In parallel, by enzyme-linked immunosorbent assay both SIC and rSIC bound strongly to C5b67 and C5b678 complexes and to a lesser extent C5b-9, but only weakly to individual complement components. The implications of these data for virulence of SIC-positive streptococci are discussed, in light of the fact that Gram-positive organisms are already protected against complement lysis by the presence of their peptidoglycan cell walls. We speculate that MAC inhibition may not be the sole function of SIC.
Figures

References
-
- Johnsson E, Berggård K, Kotarsky H, Hellwage J, Zipfel PF, Sjöbring U, Lindahl G. Role of the hypervariable region in streptococcal M proteins: binding of a human complement inhibitor. J Immunol. 1998;161:4894–901. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

