Prolonged gray matter disease without demyelination caused by Theiler's murine encephalomyelitis virus with a mutation in VP2 puff B
- PMID: 11462022
- PMCID: PMC114985
- DOI: 10.1128/JVI.75.16.7494-7505.2001
Prolonged gray matter disease without demyelination caused by Theiler's murine encephalomyelitis virus with a mutation in VP2 puff B
Abstract
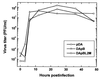
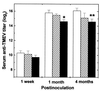
Theiler's murine encephalomyelitis virus (TMEV) is divided into two subgroups based on neurovirulence. During the acute phase, DA virus infects cells in the gray matter of the central nervous system (CNS). Throughout the chronic phase, DA virus infects glial cells in the white matter, causing demyelinating disease. Although GDVII virus also infects neurons in the gray matter, infected mice developed a severe polioencephalomyelitis, and no virus is detected in the white matter or other areas in the CNS in rare survivors. Several sequence differences between the two viruses are located in VP2 puff B and VP1 loop II, which are located near each other, close to the proposed receptor binding site. We constructed a DA virus mutant, DApBL2M, which has the VP1 loop II of GDVII virus and a mutation at position 171 in VP2 puff B. While DApBL2M virus replicated less efficiently than DA virus during the acute phase, DApBL2M-induced acute polioencephalitis was comparable to that in DA virus infection. Interestingly, during the chronic phase, DApBL2M caused prolonged gray matter disease in the brain without white matter involvement in the spinal cord. This is opposite what is observed during wild-type DA virus infection. Our study is the first to demonstrate that conformational differences via interaction of VP2 puff B and VP1 loop II between GDVII and DA viruses can play an important role in making the transition of infection from the gray matter in the brain to the spinal cord white matter during TMEV infection.
Figures





Similar articles
-
The effect of Theiler's murine encephalomyelitis virus (TMEV) VP1 carboxyl region on the virus-induced central nervous system disease.J Neurovirol. 1995 Mar;1(1):101-10. doi: 10.3109/13550289509111014. J Neurovirol. 1995. PMID: 9222346
-
Replacement of loop II of VP1 of the DA strain with loop II of the GDVII strain of Theiler's murine encephalomyelitis virus alters neurovirulence, viral persistence, and demyelination.J Virol. 1998 Sep;72(9):7557-62. doi: 10.1128/JVI.72.9.7557-7562.1998. J Virol. 1998. PMID: 9696853 Free PMC article.
-
Role of VP2 amino acid 141 in tropism of Theiler's virus within the central nervous system.J Virol. 1996 Nov;70(11):8213-7. doi: 10.1128/JVI.70.11.8213-8217.1996. J Virol. 1996. PMID: 8892956 Free PMC article.
-
Theiler's murine encephalomyelitis virus (TMEV): the role of a small out-of-frame protein in viral persistence and demyelination.Jpn J Infect Dis. 1999 Dec;52(6):228-33. Jpn J Infect Dis. 1999. PMID: 10738359 Review.
-
Molecular pathogenesis of Theiler's murine encephalomyelitis virus-induced demyelinating disease in mice.Intervirology. 1997;40(2-3):143-52. doi: 10.1159/000150541. Intervirology. 1997. PMID: 9450231 Review.
Cited by
-
Injection of the sciatic nerve with TMEV: a new model for peripheral nerve demyelination.Virology. 2007 Mar 1;359(1):233-42. doi: 10.1016/j.virol.2006.09.009. Epub 2006 Oct 9. Virology. 2007. PMID: 17028060 Free PMC article.
-
Contrasting roles for axonal degeneration in an autoimmune versus viral model of multiple sclerosis: When can axonal injury be beneficial?Am J Pathol. 2007 Jan;170(1):214-26. doi: 10.2353/ajpath.2007.060683. Am J Pathol. 2007. PMID: 17200195 Free PMC article.
-
Positive modulation of mGluR5 attenuates seizures and reduces TNF-α+ macrophages and microglia in the brain in a murine model of virus-induced temporal lobe epilepsy.Exp Neurol. 2019 Jan;311:194-204. doi: 10.1016/j.expneurol.2018.10.006. Epub 2018 Oct 11. Exp Neurol. 2019. PMID: 30316834 Free PMC article.
-
Th17-biased RORγt transgenic mice become susceptible to a viral model for multiple sclerosis.Brain Behav Immun. 2015 Jan;43:86-97. doi: 10.1016/j.bbi.2014.07.008. Epub 2014 Jul 18. Brain Behav Immun. 2015. PMID: 25046854 Free PMC article.
-
Infiltrating macrophages are key to the development of seizures following virus infection.J Virol. 2013 Feb;87(3):1849-60. doi: 10.1128/JVI.02747-12. Epub 2012 Dec 12. J Virol. 2013. PMID: 23236075 Free PMC article.
References
-
- Balashov K, Weiner H L. Multiple sclerosis. In: Eisenbarth G S, editor. Medical intelligence unit 13. Endocrine and organ specific autoimmunity. Austin, Tex: R. G. Landes Company; 1999. pp. 195–212.
-
- Burton G F, Masuda A, Heath S L, Smith B A, Tew J G, Szakal A K. Follicular dendritic cells (FDC) in retroviral infection: host/pathogen perspectives. Immunol Rev. 1997;156:185–197. - PubMed
-
- Dal Canto M C, Kim B S, Miller S D, Melvold R W. Theiler's murine encephalomyelitis virus (TMEV)-induced demyelination: a model for human multiple sclerosis. Methods. 1996;10:453–461. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

