Progesterone blockade of estrogen activation of mu-opioid receptors regulates reproductive behavior
- PMID: 11466444
- PMCID: PMC6762652
- DOI: 10.1523/JNEUROSCI.21-15-05723.2001
Progesterone blockade of estrogen activation of mu-opioid receptors regulates reproductive behavior
Abstract
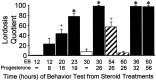
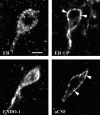
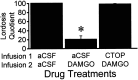
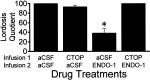
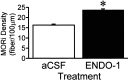
The mu-opioid receptor (MOR), a G-protein-coupled receptor, is internalized after endogenous agonist binding. Although receptor activation and internalization are separate events, internalization is a good assay for activation because endogenous opioid peptides all induce internalization. Estrogen treatment of ovariectomized rats induces MOR internalization, providing a neurochemical signature of estrogen activation of the medial preoptic nucleus. MOR activation appears to be the mechanism via which estrogen acts in the medial preoptic area to prevent the display of female reproductive behavior during the first 20-24 hr after estrogen treatment. Naltrexone, an alkaloid universal opioid receptor antagonist, prevented MOR internalization, suggesting that estrogen induces the release of endogenous opioid peptides that in turn activate the MOR. Enkephalins and beta-endorphin are nonselective endogenous MOR ligands. The most selective endogenous MOR ligands are the endomorphins. Infusions of selective MOR agonists, H-Tyr-d-Ala-Gly-N-Met-Phe-glycinol-enkephalin (DAMGO) or endomorphin-1, into the medial preoptic nucleus attenuated lordosis, and their effects were blocked with the MOR antagonist H-d-Phe-Cys-Tyr-d-Trp-Orn-Thr-Pen-Thr-NH(2) (CTOP). Infusion of endomorphin-1 internalized MOR. To determine whether progestin also acts via the MOR system to facilitate reproductive behavior, ovariectomized rats were primed with 17beta-estradiol and progesterone. Progestin facilitation of lordosis was correlated with a reduction of estrogen-induced MOR internalization. Progestin reversed estrogen-induced MOR internalization, suggesting that progesterone blocked estrogen-induced endogenous opioid release, relieving estrogen inhibition and facilitating lordosis. These results indicate a central role of MOR in the mediation of sex steroid activation of the CNS to regulate female reproductive behavior.
Figures


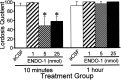
References
-
- Babcock AM, Block GJ, Micevych PE. Injections of cholecystokinin into the ventromedial hypothalamic nucleus inhibit lordosis behavior in the rat. Physiol Behav. 1988;43:195–199. - PubMed
-
- Beach F. Hormones and behavior. Hoeber; New York: 1948.
-
- Eckersell CB, Micevych PE. Opiate receptors modulate estrogen-induced cholecystokinin and tachykinin but not enkephalin mRNA levels in the limbic system and hypothalamus. Neuroscience. 1997;80:473–485. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
