Supersensitivity to anandamide and enhanced endogenous cannabinoid signaling in mice lacking fatty acid amide hydrolase
- PMID: 11470906
- PMCID: PMC55427
- DOI: 10.1073/pnas.161191698
Supersensitivity to anandamide and enhanced endogenous cannabinoid signaling in mice lacking fatty acid amide hydrolase
Abstract
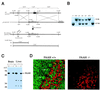
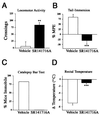
The medicinal properties of marijuana have been recognized for centuries, but clinical and societal acceptance of this drug of abuse as a potential therapeutic agent remains fiercely debated. An attractive alternative to marijuana-based therapeutics would be to target the molecular pathways that mediate the effects of this drug. To date, these neural signaling pathways have been shown to comprise a cannabinoid receptor (CB(1)) that binds the active constituent of marijuana, tetrahydrocannabinol (THC), and a postulated endogenous CB(1) ligand anandamide. Although anandamide binds and activates the CB(1) receptor in vitro, this compound induces only weak and transient cannabinoid behavioral effects in vivo, possibly a result of its rapid catabolism. Here we show that mice lacking the enzyme fatty acid amide hydrolase (FAAH(-/-)) are severely impaired in their ability to degrade anandamide and when treated with this compound, exhibit an array of intense CB(1)-dependent behavioral responses, including hypomotility, analgesia, catalepsy, and hypothermia. FAAH(-/-)-mice possess 15-fold augmented endogenous brain levels of anandamide and display reduced pain sensation that is reversed by the CB(1) antagonist SR141716A. Collectively, these results indicate that FAAH is a key regulator of anandamide signaling in vivo, setting an endogenous cannabinoid tone that modulates pain perception. FAAH may therefore represent an attractive pharmaceutical target for the treatment of pain and neuropsychiatric disorders.
Figures

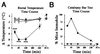

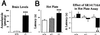
References
-
- Pop E. Curr Opin Chem Biol. 1999;3:418–425. - PubMed
-
- Piomelli D, Giuffrida A, Calignano A, Rodriguez de Fonseca F. Trends Pharmacol Sci. 2000;21:218–224. - PubMed
-
- Martin B R, Mechoulam R, Razdan R K. Life Sci. 1999;65:573–595. - PubMed
-
- Mechoulam R. Cannabinoids as Therapeutic Agents. Boca Raton, FL: CRC; 1986. pp. 1–19.
-
- Pertwee R G. Pharmacol Ther. 1997;74:129–180. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

