Neuronal Ca(V)1.3alpha(1) L-type channels activate at relatively hyperpolarized membrane potentials and are incompletely inhibited by dihydropyridines
- PMID: 11487617
- PMCID: PMC6763157
- DOI: 10.1523/JNEUROSCI.21-16-05944.2001
Neuronal Ca(V)1.3alpha(1) L-type channels activate at relatively hyperpolarized membrane potentials and are incompletely inhibited by dihydropyridines
Abstract
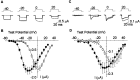
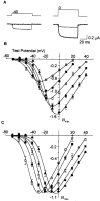
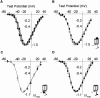
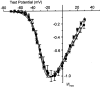
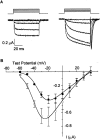
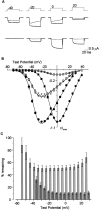
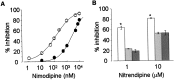
L-type calcium channels regulate a diverse array of cellular functions within excitable cells. Of the four molecularly defined subclasses of L-type Ca channels, two are expressed ubiquitously in the mammalian nervous system (Ca(V)1.2alpha(1) and Ca(V)1.3alpha(1)). Despite diversity at the molecular level, neuronal L-type channels are generally assumed to be functionally and pharmacologically similar, i.e., high-voltage activated and highly sensitive to dihydropyridines. We now show that Ca(V)1.3alpha(1) L-type channels activate at membrane potentials approximately 25 mV more hyperpolarized, compared with Ca(V)1.2alpha(1). This unusually negative activation threshold for Ca(V)1.3alpha(1) channels is independent of the specific auxiliary subunits coexpressed, of alternative splicing in domains I-II, IVS3-IVS4, and the C terminus, and of the expression system. The use of high concentrations of extracellular divalent cations has possibly obscured the unique voltage-dependent properties of Ca(V)1.3alpha(1) in certain previous studies. We also demonstrate that Ca(V)1.3alpha(1) channels are pharmacologically distinct from Ca(V)1.2alpha(1). The IC(50) for nimodipine block of Ca(V)1.3alpha(1) L-type calcium channel currents is 2.7 +/- 0.3 microm, a value 20-fold higher than the concentration required to block Ca(V)1.2alpha(1). The relatively low sensitivity of the Ca(V)1.3alpha(1) subunit to inhibition by dihydropyridine is unaffected by alternative splicing in the IVS3-IVS4 linker. Our results suggest that functional and pharmacological criteria used commonly to distinguish among different Ca currents greatly underestimate the biological importance of L-type channels in cells expressing Ca(v)1.3alpha(1).
Figures
References
-
- Ashcroft FM, Proks P, Smith PA, Ammala C, Bokvist K, Rorsman P. Stimulus-secretion coupling in pancreatic beta cells. J Cell Biochem. 1994;55:54–65. - PubMed
-
- Beam KG, Tanabe T, Numa S. Structure, function, and regulation of the skeletal muscle dihydropyridine receptor. Ann NY Acad Sci. 1989;560:127–137. - PubMed
-
- Bean BP. Classes of calcium channels in vertebrate cells. Annu Rev Physiol. 1989;51:367–384. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
