Cortical slow oscillatory activity is reflected in the membrane potential and spike trains of striatal neurons in rats with chronic nigrostriatal lesions
- PMID: 11487667
- PMCID: PMC6763136
- DOI: 10.1523/JNEUROSCI.21-16-06430.2001
Cortical slow oscillatory activity is reflected in the membrane potential and spike trains of striatal neurons in rats with chronic nigrostriatal lesions
Abstract
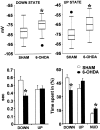
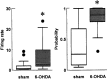
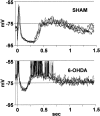
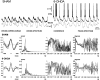
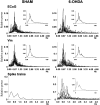
Neurons in the basal ganglia output nuclei display rhythmic burst firing after chronic nigrostriatal lesions. The thalamocortical network is a strong endogenous generator of oscillatory activity, and the striatum receives a massive projection from the cerebral cortex. Actually, the membrane potential of striatal projection neurons displays periodic shifts between a very negative resting potential (down state) and depolarizing plateaus (up states) during which they can fire action potentials. We hypothesized that an increased excitability of striatal neurons may allow transmission of cortical slow rhythms through the striatum to the remaining basal ganglia in experimental parkinsonism. In vivo intracellular recordings revealed that striatal projection neurons from rats with chronic nigrostriatal lesions had a more depolarized membrane potential during both the down and up states and an increased firing probability during the up events. Furthermore, lesioned rats had significantly fewer silent neurons than control rats. Simultaneous recordings of the frontal electrocorticogram and membrane potential of striatal projection neurons revealed that the signals were oscillating synchronously in the frequency range 0.4-2 Hz, both in control rats and rats with chronic nigrostriatal lesions. Spreading of the slow cortical rhythm is limited by the very low firing probability of control rat neurons, but a slow oscillation is well reflected in spike trains of approximately 60% of lesioned rat neurons. These findings provide in vivo evidence for a role of dopamine in controlling the flow of cortical activity through the striatum and may be of outstanding relevance for understanding the pathophysiology of Parkinson's disease.
Figures


References
-
- Albin RL, Young AB, Penney JB. The functional anatomy of basal ganglia disorders. Trends Neurosci. 1989;12:366–375. - PubMed
-
- Amzica F, Steriade M. Short- and long-range neuronal synchronization of the slow (<1 Hz) cortical oscillation. J Neurophysiol. 1995;73:604–622. - PubMed
-
- Amzica F, Steriade M. Cellular substrates and laminar profile of sleep K-complex. Neuroscience. 1998;82:671–686. - PubMed
-
- Arbuthnott GW. Spontaneous activity of single units in the striatum after unilateral destruction of the dopamine input. J Physiol (Lond) 1974;239:121P–122P. - PubMed
-
- Bergman H, Wichmann T, Karmon B, DeLong MR. The primate subthalamic nucleus. II. Neuronal activity in the MPTP model of parkinsonism. J Neurophysiol. 1994;72:507–520. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
