Examining human T-lymphotropic virus type 1 infection and replication by cell-free infection with recombinant virus vectors
- PMID: 11507191
- PMCID: PMC115091
- DOI: 10.1128/jvi.75.18.8461-8468.2001
Examining human T-lymphotropic virus type 1 infection and replication by cell-free infection with recombinant virus vectors
Abstract
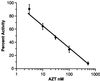
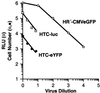
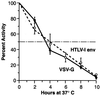
A sensitive and quantitative cell-free infection assay, utilizing recombinant human T-cell leukemia virus type 1 (HTLV-1)-based vectors, was developed in order to analyze early events in the virus replication cycle. Previous difficulties with the low infectivity and restricted expression of the virus have prevented a clear understanding of these events. Virus stocks were generated by transfecting cells with three plasmids: (i) a packaging plasmid encoding HTLV-1 structural and regulatory proteins, (ii) an HTLV-1 transfer vector containing either firefly luciferase or enhanced yellow fluorescent protein genes, and (iii) an envelope expression plasmid. Single-round infections were initiated by exposing target cells to filtered supernatants and quantified by assaying for luciferase activity in cell extracts or by enumerating transduced cells by flow cytometry. Transduction was dependent on reverse transcription and integration of the recombinant virus genome, as shown by the effects of the reverse transcriptase inhibitor 3'-azido-3'-deoxythymidine (AZT) and by mutation of the integrase gene in the packaging vector, respectively. The 50% inhibitory concentration of AZT was determined to be 30 nM in this HTLV-1 replication system. The stability of HTLV-1 particles, pseudotyped with either vesicular stomatitis virus G protein or HTLV-1 envelope, was typical of retroviruses, exhibiting a half-life of approximately 3.5 h at 37 degrees C. The specific infectivity of recombinant HTLV-1 virions was at least 3 orders of magnitude lower than that of analogous HIV-1 particles, though both were pseudotyped with the same envelope. Thus, the low infectivity of HTLV-1 is determined in large part by properties of the core particle and by the efficiency of postentry processes.
Figures


References
-
- Boucher C A, O'Sullivan E, Mulder J W, Ramautarsing C, Kellam P, Darby G, Lange J M, Goudsmit J, Larder B A. Ordered appearance of zidovudine resistance mutations during treatment of 18 human immunodeficiency virus-positive subjects. J Infect Dis. 1992;165:105–110. - PubMed
-
- Clapham P, Nagy K, Cheingsong-Popov R, Exley M, Weiss R A. Productive infection and cell-free transmission of human T-cell leukemia virus in a nonlymphoid cell line. Science. 1983;222:1125–1127. - PubMed
-
- de Rossi A, Aldovini A, Franchini G, Mann D, Gallo R C, Wong-Staal F. Clonal selection of T lymphocytes infected by cell-free human T-cell leukemia/lymphoma virus type I: parameters of virus integration and expression. Virology. 1985;143:640–645. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

