Recruitment of the mitochondrial-dependent apoptotic pathway in amyotrophic lateral sclerosis
- PMID: 11517246
- PMCID: PMC6763092
- DOI: 10.1523/JNEUROSCI.21-17-06569.2001
Recruitment of the mitochondrial-dependent apoptotic pathway in amyotrophic lateral sclerosis
Abstract
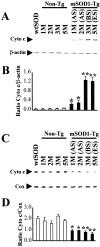
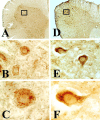
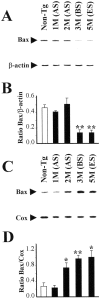
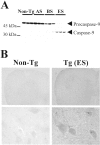
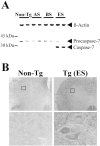
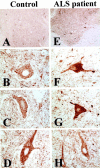
Molecular mechanisms of apoptosis may participate in motor neuron degeneration produced by mutant superoxide dismutase-1 (mSOD1), the only proven cause of amyotrophic lateral sclerosis (ALS). Consistent with this, here we show that the proapoptotic protein Bax translocates from the cytosol to the mitochondria, whereas cytochrome c translocates from the mitochondria to the cytosol in spinal cords of transgenic mSOD1 mice during the progression of the disease. Concomitantly, caspase-9 is activated in the spinal cord of transgenic mSOD1 mice. Only in end-stage transgenic mSOD1 mice is the downstream caspase-7 activated and the inhibitor of apoptosis, XIAP, cleaved. These results indicate a sequential recruitment of molecular elements of the mitochondrial-dependent apoptotic pathway in transgenic mSOD1 mice. We also provide immunohistochemical evidence that cytochrome c translocation occurs in the spinal cord of sporadic ALS patients. Collectively, these data suggest that the mitochondrial-dependent apoptotic pathway may contribute to the demise of motor neurons in ALS and that targeting key molecules of this cascade may prove to be neuroprotective.
Figures

References
-
- Adams JH, Corsellis JAN, Duchen LW. Greenfield's neuropathology. Butler and Tanner; London: 1984.
-
- Almer G, Vukosavic S, Romero N, Przedborski S. Inducible nitric oxide synthase upregulation in a transgenic mouse model of familial amyotrophic lateral sclerosis. J Neurochem. 1999;72:2415–2425. - PubMed
-
- Brown RH., Jr Superoxide dismutase in familial amyotrophic lateral sclerosis: models for gain of function. Curr Opin Neurobiol. 1995;5:841–846. - PubMed
-
- Bruijn LI, Cleveland DW. Mechanisms of selective motor neuron death in ALS: insights from transgenic mouse models of motor neuron disease. Neuropathol Appl Neurobiol. 1996;22:373–387. - PubMed
-
- Dal Canto MC, Gurney ME. Neuropathological changes in two lines of mice carrying a transgene for mutant human Cu,Zn SOD, and in mice overexpressing wild type human SOD: a model of familial amyotrophic lateral sclerosis (FALS). Brain Res. 1995;676:25–40. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous
