Receptor heterodimerization: essential mechanism for platelet-derived growth factor-induced epidermal growth factor receptor transactivation
- PMID: 11533228
- PMCID: PMC99786
- DOI: 10.1128/MCB.21.19.6387-6394.2001
Receptor heterodimerization: essential mechanism for platelet-derived growth factor-induced epidermal growth factor receptor transactivation
Abstract
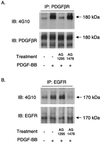
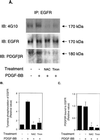
Previous studies showed that the epidermal growth factor receptor (EGFR) can be transactivated by platelet-derived growth factor (PDGF) stimulation and that EGFR transactivation is required for PDGF-stimulated cell migration. To investigate the mechanism for cross talk between the PDGF beta receptor (PDGFbetaR) and the EGFR, we stimulated rat aortic vascular smooth muscle cells (VSMC) with 20 ng of PDGF/ml. Transactivation of the EGFR, defined by receptor tyrosine phosphorylation, occurred with the same time course as PDGFbetaR activation. Basal formation of PDGFbetaR-EGFR heterodimers was shown by coimmunoprecipitation studies, and interestingly, disruption of this receptor heterodimer abolished EGFR transactivation. Breakdown of the heterodimer was observed when VSMC were pretreated with antioxidants or with a Src family kinase inhibitor. Disruption of heterodimers decreased ERK1 and ERK2 activation by PDGF. Although PDGF-induced PDGFbetaR activation was abolished after pretreatment with 1 microM AG1295 (a specific PDGF receptor kinase inhibitor), EGFR transactivation was still observed, indicating that PDGFbetaR kinase activity is not required. In conclusion, our data demonstrate that the PDGFbetaR and the EGFR form PDGFbetaR-EGFR heterodimers basally, and we suggest that heterodimers represent a novel signaling complex which plays an important role in PDGF signal transduction.
Figures



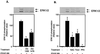
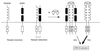
Similar articles
-
Sphingosine 1-phosphate transactivates the platelet-derived growth factor beta receptor and epidermal growth factor receptor in vascular smooth muscle cells.Circ Res. 2004 Apr 30;94(8):1050-8. doi: 10.1161/01.RES.0000126404.41421.BE. Epub 2004 Mar 25. Circ Res. 2004. PMID: 15044318
-
P2Y receptor-mediated stimulation of Müller glial cell DNA synthesis: dependence on EGF and PDGF receptor transactivation.Invest Ophthalmol Vis Sci. 2003 Mar;44(3):1211-20. doi: 10.1167/iovs.02-0260. Invest Ophthalmol Vis Sci. 2003. PMID: 12601051
-
Phosphoinositide-dependent kinase 1 and p21-activated protein kinase mediate reactive oxygen species-dependent regulation of platelet-derived growth factor-induced smooth muscle cell migration.Circ Res. 2004 May 14;94(9):1219-26. doi: 10.1161/01.RES.0000126848.54740.4A. Epub 2004 Apr 1. Circ Res. 2004. PMID: 15059930
-
Increased angiotensin II-mediated Src signaling via epidermal growth factor receptor transactivation is associated with decreased C-terminal Src kinase activity in vascular smooth muscle cells from spontaneously hypertensive rats.Hypertension. 2002 Feb;39(2 Pt 2):479-85. doi: 10.1161/hy02t2.102909. Hypertension. 2002. PMID: 11882594
-
Angiotensin II induces transactivation of two different populations of the platelet-derived growth factor beta receptor. Key role for the p66 adaptor protein Shc.J Biol Chem. 2000 May 26;275(21):15926-32. doi: 10.1074/jbc.M909616199. J Biol Chem. 2000. PMID: 10748142
Cited by
-
Pvr regulates cyst stem cell division in the Drosophila testis niche, and has functions distinct from Egfr.Cells Dev. 2023 Mar;173:203822. doi: 10.1016/j.cdev.2022.203822. Epub 2022 Nov 16. Cells Dev. 2023. PMID: 36400422 Free PMC article.
-
Role of Epidermal Growth Factor Receptor (EGFR) and Its Ligands in Kidney Inflammation and Damage.Mediators Inflamm. 2018 Dec 23;2018:8739473. doi: 10.1155/2018/8739473. eCollection 2018. Mediators Inflamm. 2018. PMID: 30670929 Free PMC article. Review.
-
PDGF-induced proliferation in human arterial and venous smooth muscle cells: molecular basis for differential effects of PDGF isoforms.J Cell Biochem. 2011 Jan;112(1):289-98. doi: 10.1002/jcb.22924. J Cell Biochem. 2011. PMID: 21069732 Free PMC article.
-
The Role of Platelets in the Tumor-Microenvironment and the Drug Resistance of Cancer Cells.Cancers (Basel). 2019 Feb 19;11(2):240. doi: 10.3390/cancers11020240. Cancers (Basel). 2019. PMID: 30791448 Free PMC article. Review.
-
One-pot synthesis of heterodimeric agonists that activate the canonical Wnt signaling pathway.Chem Commun (Camb). 2020 Mar 26;56(25):3685-3688. doi: 10.1039/d0cc00920b. Chem Commun (Camb). 2020. PMID: 32119023 Free PMC article.
References
-
- Abe J, Berk B C. Fyn and JAK2 mediate Ras activation by reactive oxygen species. J Biol Chem. 1999;274:21003–21010. - PubMed
-
- Abe J, Okuda M, Huang Q, Yoshizumi M, Berk B C. Reactive oxygen species activate p90 ribosomal S6 kinase via Fyn and Ras. J Biol Chem. 2000;275:1739–1748. - PubMed
-
- Abe J, Takahashi M, Ishida M, Lee J D, Berk B C. c-Src is required for oxidative stress-mediated activation of big mitogen-activated protein kinase. 1. J Biol Chem. 1997;272:20389–20394. - PubMed
-
- Bae Y S, Kang S W, Seo M S, Baines I C, Tekle E, Chock P B, Rhee S G. Epidermal growth factor (EGF)-induced generation of hydrogen peroxide. Role in EGF receptor-mediated tyrosine phosphorylation. J Biol Chem. 1997;272:217–221. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
