5-hydroxytryptamine strongly inhibits fluid secretion in guinea pig pancreatic duct cells
- PMID: 11544281
- PMCID: PMC209377
- DOI: 10.1172/JCI12312
5-hydroxytryptamine strongly inhibits fluid secretion in guinea pig pancreatic duct cells
Abstract
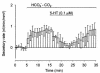
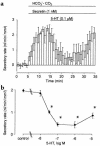
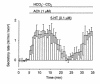
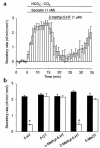
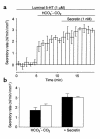
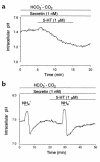
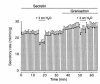
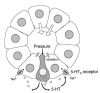
We studied the distribution of 5-hydroxytryptamine- (5-HT-) containing cells in the guinea pig pancreas and examined the effects of 5-HT on fluid secretion by interlobular pancreatic ducts. The 5-HT-immunoreactive cells with morphological characteristics of enterochromaffin (EC) cells were scattered throughout the duct system and were enriched in islets of Langerhans. The fluid secretory rate in the isolated interlobular ducts was measured by videomicroscopy. Basolateral applications of 5-HT strongly but reversibly reduced HCO(3)-dependent, as well as secretin- and acetylcholine- (ACh-) stimulated, fluid secretion, whereas 5-HT applied into the lumen had no such effects. Secretin-stimulated fluid secretion could be inhibited by a 5-HT(3) receptor agonist, but not by agonists of the 5-HT(1), 5-HT(2), or 5-HT(4) receptors. Under the stimulation with secretin, 5-HT decreased the intracellular pH (pH(i)) and reduced the rate of pH(i) recovery after acid loading with NH(4)(+), suggesting that 5-HT inhibits the intracellular accumulation of HCO3(-). The elevation of intraductal pressure in vivo reduced secretin-stimulated fluid secretion, an effect that could be attenuated by a 5-HT(3) receptor antagonist. Thus, 5-HT, acting through basolateral 5-HT(3) receptors, strongly inhibits spontaneous, secretin-, and ACh-stimulated fluid secretion by guinea pig pancreatic ducts. 5-HT released from pancreatic ductal EC cells on elevation of the intraductal pressure may regulate fluid secretion of neighboring duct cells in a paracrine fashion.
Figures

Similar articles
-
Ethanol induces fluid hypersecretion from guinea-pig pancreatic duct cells.J Physiol. 2003 Sep 15;551(Pt 3):917-26. doi: 10.1113/jphysiol.2003.048827. Epub 2003 Jul 7. J Physiol. 2003. PMID: 12847207 Free PMC article.
-
Basolateral anion transport mechanisms underlying fluid secretion by mouse, rat and guinea-pig pancreatic ducts.J Physiol. 2004 Apr 15;556(Pt 2):415-28. doi: 10.1113/jphysiol.2004.061762. Epub 2004 Feb 20. J Physiol. 2004. PMID: 14978209 Free PMC article.
-
Serotonin and pancreatic duct function.J Korean Med Sci. 2000 Aug;15 Suppl(Suppl):S27-8. doi: 10.3346/jkms.2000.15.S.S27. J Korean Med Sci. 2000. PMID: 10981503 Free PMC article.
-
Secretin-dependent HCO3- secretion from pancreas and liver.J Intern Med Suppl. 1990;732:47-51. J Intern Med Suppl. 1990. PMID: 2200415 Review.
-
The inhibitory pathways of pancreatic ductal bicarbonate secretion.Int J Biochem Cell Biol. 2007;39(1):25-30. doi: 10.1016/j.biocel.2006.07.011. Epub 2006 Aug 18. Int J Biochem Cell Biol. 2007. PMID: 16996776 Review.
Cited by
-
Intracellular Ca2+ Signalling in the Pathogenesis of Acute Pancreatitis: Recent Advances and Translational Perspectives.Int J Mol Sci. 2020 Jun 3;21(11):4005. doi: 10.3390/ijms21114005. Int J Mol Sci. 2020. PMID: 32503336 Free PMC article. Review.
-
Altered serotonin physiology in human breast cancers favors paradoxical growth and cell survival.Breast Cancer Res. 2009;11(6):R81. doi: 10.1186/bcr2448. Epub 2009 Nov 10. Breast Cancer Res. 2009. PMID: 19903352 Free PMC article.
-
Streptozotocin-Induced Diabetes Causes Changes in Serotonin-Positive Neurons in the Small Intestine in Pig Model.Int J Mol Sci. 2022 Apr 20;23(9):4564. doi: 10.3390/ijms23094564. Int J Mol Sci. 2022. PMID: 35562954 Free PMC article.
-
Intraluminal volume homeostasis: A common sertonergic mechanism among diverse epithelia.Commun Integr Biol. 2011 Sep;4(5):532-7. doi: 10.4161/cib.16492. Epub 2011 Sep 1. Commun Integr Biol. 2011. PMID: 22046455 Free PMC article.
-
Ethanol induces fluid hypersecretion from guinea-pig pancreatic duct cells.J Physiol. 2003 Sep 15;551(Pt 3):917-26. doi: 10.1113/jphysiol.2003.048827. Epub 2003 Jul 7. J Physiol. 2003. PMID: 12847207 Free PMC article.
References
-
- Sjolund K, Sanden G, Hakanson R, Sundler F. Endocrine cells in human intestine: an immunocytochemical study. Gastroenterology. 1983;85:1120–1130. - PubMed
-
- Gershon, M.D., Kirchgessner, A.L., and Wade, P.R. 1994. Functional anatomy of the enteric nervous system. In Physiology of the gastrointestinal tract. L.R. Johnson, editor. Raven Press. New York, New York, USA. 381–422.
-
- Nilsson O, Ahlman H, Geffard M, Dahlström A, Ericson LE. Bipolarity of duodenal enterochromaffin cells in the rat. Cell Tissue Res. 1987;248:49–54. - PubMed
-
- Hubel KA, Renquist KS, Varley G. Secretory reflexes in ileum and jejunum: absence of remote effects. J Auton Nerv Syst. 1991;35:53–62. - PubMed

