An antagonist IL-15/Fc protein prevents costimulation blockade-resistant rejection
- PMID: 11544341
- PMCID: PMC3806296
- DOI: 10.4049/jimmunol.167.6.3478
An antagonist IL-15/Fc protein prevents costimulation blockade-resistant rejection
Abstract
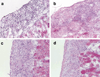
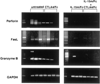
IL-15 is a powerful T cell growth factor (TCGF) with particular importance for the maintenance of CD8(+) T cells. Because costimulation blockade does not result in universal tolerance, we hypothesized that "escape" from costimulation blockade might represent a CD8(+) and IL-15/IL-15R(+)-dependent process. For this analysis, we have used an IL-15 mutant/Fcgamma2a protein, a potentially cytolytic protein that is also a high-affinity receptor site specific antagonist for the IL-15Ralpha receptor protein, as a therapeutic agent. The IL-15-related fusion protein was used as monotherapy or in combination with CTLA4/Fc in murine islet allograft models. As monotherapies, CTLA4/Fc and an IL-15 mutant/Fcgamma2a were comparably effective in a semiallogeneic model system, and combined treatment with IL-15 mutant/Fcgamma2a plus CTLA4/Fc produced universal permanent engraftment. In a fully MHC-mismatched strain combination known to be refractory to costimulation blockade treatment, combined treatment with both fusion proteins proved to be highly effective; >70% of recipients were tolerized. The analysis revealed that the IL-15 mutant/Fc treatment confers partial protection from both CD4(+) and CD8(+) T cell graft infiltration. In rejections occurring despite CTLA4/Fc treatment, concomitant treatment with the IL-15 mutant/Fcgamma2a protein blocked a CD8(+) T cell-dominated rejection processes. This protection was linked to a blunted proliferative response of alloreactive T cells as well silencing of CTL-related gene expression events. Hence, we have demonstrated that targeting the IL-15/IL-15R pathway represents a new and potent strategy to prevent costimulation blockade-resistant CD8(+) T cell-driven rejection.
Figures



Similar articles
-
CD8(+) T cells resistant to costimulatory blockade are controlled by an antagonist interleukin-15/Fc protein.Transplantation. 2006 Dec 15;82(11):1510-7. doi: 10.1097/01.tp.0000243168.53126.d2. Transplantation. 2006. PMID: 17164724 Free PMC article.
-
Addition of an IL-15 mutant/FCgamma2A antagonist protein protects islet allografts from rejection overriding costimulation blockade.Transplant Proc. 2002 May;34(3):745-7. doi: 10.1016/s0041-1345(01)02900-1. Transplant Proc. 2002. PMID: 12034169 Free PMC article. No abstract available.
-
The role of the IL-2 pathway in costimulation blockade-resistant rejection of allografts.J Immunol. 2002 Feb 1;168(3):1123-30. doi: 10.4049/jimmunol.168.3.1123. J Immunol. 2002. PMID: 11801646
-
The mosaic of immunosuppressive drugs.Mol Immunol. 2003 Jul;39(17-18):1073-7. doi: 10.1016/s0161-5890(03)00075-0. Mol Immunol. 2003. PMID: 12835079 Review.
-
Transition from preclinical to clinical application of CTLA4-Ig co-stimulation blockage in beta-cell replacement therapy.Transplant Rev (Orlando). 2025 Apr;39(2):100913. doi: 10.1016/j.trre.2025.100913. Epub 2025 Mar 2. Transplant Rev (Orlando). 2025. PMID: 40048867 Review.
Cited by
-
An antagonist mutant IL-15/Fc promotes transplant tolerance.Transplantation. 2006 Jan 15;81(1):109-16. doi: 10.1097/01.tp.0000188139.11931.98. Transplantation. 2006. PMID: 16421485 Free PMC article.
-
Implications of glycosylation for the development of selected cytokines and their derivatives for medical use.Biotechnol Adv. 2024 Dec;77:108467. doi: 10.1016/j.biotechadv.2024.108467. Epub 2024 Oct 22. Biotechnol Adv. 2024. PMID: 39447666 Free PMC article. Review.
-
Targeting cytokines beyond tumor necrosis factor-alpha and interleukin-1 in rheumatoid arthritis.Curr Rheumatol Rep. 2004 Oct;6(5):336-42. doi: 10.1007/s11926-004-0007-2. Curr Rheumatol Rep. 2004. PMID: 15355745 Review.
-
Pig embryonic pancreatic tissue as a source for transplantation in diabetes: transient treatment with anti-LFA1, anti-CD48, and FTY720 enables long-term graft maintenance in mice with only mild ongoing immunosuppression.Diabetes. 2009 Jul;58(7):1585-94. doi: 10.2337/db09-0112. Epub 2009 Apr 28. Diabetes. 2009. PMID: 19401429 Free PMC article.
-
T-cell-directed treatment strategies for Type 1 diabetes and the confounding role of inflammation.Immunotherapy. 2010 Jul;2(4):431-6. doi: 10.2217/imt.10.40. Immunotherapy. 2010. PMID: 20635995 Free PMC article. No abstract available.
References
-
- June CH, Bluestone JA, Nadler LM, Thompson CB. The B7 and CD28 receptor families. Immunol. Today. 1994;15:321. - PubMed
-
- Lenschow DJ, Walunas TL, Bluestone JA. CD28/B7 system of T-cell costimulation. Annu. Rev. Immunol. 1996;14:233. - PubMed
-
- Grewal IS, Flavell RA. CD40 and CD154 in cell mediated immunity. Annu. Rev. Immunol. 1998;16:111. R. A. - PubMed
-
- Marengere LE, Waterhouse P, Duncan GS, Mittrucker H-W, Feng GS, Mak TW. Regulation of T cell receptor signaling by tyrosine phosphatase in association with CTLA-4. Science. 1996;272:1170. - PubMed
-
- Lenschow DJ, Zeng Y, Thistlethwaite JR, Montag A, Brady W, Gibson MG, Linsley PS, Bluestone JA. Long-term survival of xenogeneic pancreatic islet grafts induced by CTLA4Ig. Science. 1992;257:789. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

