D box and KEN box motifs in budding yeast Hsl1p are required for APC-mediated degradation and direct binding to Cdc20p and Cdh1p
- PMID: 11562348
- PMCID: PMC312781
- DOI: 10.1101/gad.917901
D box and KEN box motifs in budding yeast Hsl1p are required for APC-mediated degradation and direct binding to Cdc20p and Cdh1p
Abstract
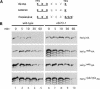
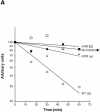
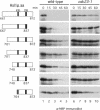
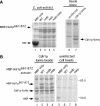
The precise order of molecular events during cell cycle progression depends upon ubiquitin-mediated proteolysis of cell cycle regulators. We demonstrated previously that Hsl1p, a protein kinase that inhibits the Swe1p protein kinase in a bud morphogenesis checkpoint, is targeted for ubiquitin-mediated turnover by the anaphase-promoting complex (APC). Here, we investigate regions of Hsl1p that are critical both for binding to the APC machinery and for APC-mediated degradation. We demonstrate that Hsl1p contains both a destruction box (D box) and a KEN box motif that are necessary for Hsl1p turnover with either APC(Cdc20) or APC(Cdh1). In coimmunoprecipitation studies, the D box of full-length Hsl1p was critical for association with Cdc20p, whereas the KEN box was important for association with Cdh1p. Fusion of a 206-amino-acid fragment of Hsl1p containing these motifs to a heterologous protein resulted in APC-dependent degradation of the fusion protein that required intact D box and KEN box motifs. Finally, this bacterially expressed Hsl1p fusion protein interacted with Cdc20p and Cdh1p either translated in vitro or expressed in and purified from insect cells. Binding to Cdc20p and Cdh1p was disrupted completely by a D box/KEN box double mutant. These results indicate that D box and KEN box motifs are important for direct binding to the APC machinery, leading to ubiquitination and subsequent protein degradation.
Figures




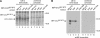
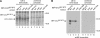
References
-
- Amon A, Irniger S, Nasmyth K. Closing the cell cycle circle in yeast: G2 cyclin proteolysis initiated at mitosis persists until the activation of G1 cyclins in the next cycle. Cell. 1994;77:1037–1050. - PubMed
-
- Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K. Current Protocols in Molecular Biology. New York: John Wiley & Sons; 1995.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
