Inhalation delivery of proteins from ethanol suspensions
- PMID: 11562495
- PMCID: PMC58690
- DOI: 10.1073/pnas.201413798
Inhalation delivery of proteins from ethanol suspensions
Abstract
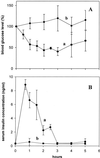
To circumvent inherent problems associated with pulmonary administration of aqueous-solution and dry-powder protein drugs, inhalation delivery of proteins from their suspensions in absolute ethanol was explored both in vitro and in vivo. Protein suspensions in ethanol of up to 9% (wt/vol) were readily aerosolized with a commercial compressor nebulizer. Experiments with enzymic proteins revealed that nebulization caused no detectable loss of catalytic activity; furthermore, enzyme suspensions in anhydrous ethanol retained their full catalytic activity for at least 3 weeks at room temperature. With the use of Zn(2+)-insulin, conditions were elaborated that produced submicron protein particles in ethanol suspensions. The latter (insulin/EtOH) afforded respirable-size aerosol particles after nebulization. A 40-min exposure of laboratory rats to 10 mg/ml insulin/EtOH aerosols resulted in a 2-fold drop in the blood glucose level and a marked rise in the serum insulin level. The bioavailability based on estimated deposited lung dose of insulin delivered by inhalation of ethanol suspension aerosols was 33% (relative to an equivalent s.c. injection), i.e., comparable to those observed in rats after inhalation administration of dry powder and aqueous solutions of insulin. Inhalation of ethanol in a relevant amount/time frame resulted in no detectable acute toxic effects on rat lungs or airways, as reflected by the absence of statistically significant inflammatory or allergic responses, damage to the alveolar/capillary barrier, and lysed and/or damaged cells.
Figures
Similar articles
-
[Standard technical specifications for methacholine chloride (Methacholine) bronchial challenge test (2023)].Zhonghua Jie He He Hu Xi Za Zhi. 2024 Feb 12;47(2):101-119. doi: 10.3760/cma.j.cn112147-20231019-00247. Zhonghua Jie He He Hu Xi Za Zhi. 2024. PMID: 38309959 Chinese.
-
NTP Toxicology and Carcinogenesis Studies of Talc (CAS No. 14807-96-6)(Non-Asbestiform) in F344/N Rats and B6C3F1 Mice (Inhalation Studies).Natl Toxicol Program Tech Rep Ser. 1993 Sep;421:1-287. Natl Toxicol Program Tech Rep Ser. 1993. PMID: 12616290
-
Inhaled insulin is better absorbed when administered as a dry powder compared to solution in the presence or absence of alkylglycosides.Pharm Res. 2006 Jan;23(1):138-47. doi: 10.1007/s11095-005-8926-9. Epub 2006 Dec 7. Pharm Res. 2006. PMID: 16320001
-
Inhalation, deposition, and fate of insulin and other therapeutic proteins.Diabetes Technol Ther. 2007 Jun;9 Suppl 1:S4-S15. doi: 10.1089/dia.2007.0228. Diabetes Technol Ther. 2007. PMID: 17563302 Review.
-
The function and performance of aqueous aerosol devices for inhalation therapy.J Pharm Pharmacol. 2016 May;68(5):556-78. doi: 10.1111/jphp.12541. Epub 2016 Apr 8. J Pharm Pharmacol. 2016. PMID: 27061412 Review.
Cited by
-
Development and Characterization of Inhaled Ethanol as a Novel Pharmacological Strategy Currently Evaluated in a Phase II Clinical Trial for Early-Stage SARS-CoV-2 Infection.Pharmaceutics. 2021 Mar 5;13(3):342. doi: 10.3390/pharmaceutics13030342. Pharmaceutics. 2021. PMID: 33808025 Free PMC article.
-
Inhalable Nanotechnology-Based Drug Delivery Systems for the Treatment of Inflammatory Lung Diseases.Pharmaceutics. 2025 Jul 9;17(7):893. doi: 10.3390/pharmaceutics17070893. Pharmaceutics. 2025. PMID: 40733101 Free PMC article. Review.
-
A green triple biocide cocktail consisting of a biocide, EDDS and methanol for the mitigation of planktonic and sessile sulfate-reducing bacteria.World J Microbiol Biotechnol. 2012 Feb;28(2):431-5. doi: 10.1007/s11274-011-0832-1. Epub 2011 Jul 8. World J Microbiol Biotechnol. 2012. PMID: 22806837
-
Inhaled agonists of soluble guanylate cyclase induce selective pulmonary vasodilation.Am J Respir Crit Care Med. 2007 Dec 1;176(11):1138-45. doi: 10.1164/rccm.200707-1121OC. Epub 2007 Sep 13. Am J Respir Crit Care Med. 2007. PMID: 17872487 Free PMC article.
-
Materials for diabetes therapeutics.Adv Healthc Mater. 2012 May;1(3):267-84. doi: 10.1002/adhm.201200037. Epub 2012 Apr 5. Adv Healthc Mater. 2012. PMID: 23184741 Free PMC article. Review.
References
-
- Thayer A M. Chem Eng News. 1998;76(32):19–31.
-
- Goddard P. Adv Drug Delivery Rev. 1991;6:103–131.
-
- Wallace B M, Lasker J S. Science. 1993;260:912–913. - PubMed
-
- Lee H J. In: Peptide-Based Drug Design. Taylor M, Amidon G, editors. Washington, DC: Am. Chem. Soc.; 1995. pp. 69–97.
-
- Burke P A. In: Handbook of Pharmaceutical Controlled Release Technology. Wise D L, editor. New York: Dekker; 2000. pp. 661–692.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

