Down-regulation of beta-catenin by activated p53
- PMID: 11564862
- PMCID: PMC99855
- DOI: 10.1128/MCB.21.20.6768-6781.2001
Down-regulation of beta-catenin by activated p53
Abstract
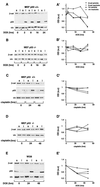
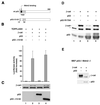
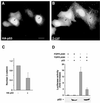
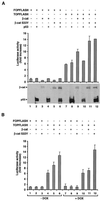
beta-Catenin is a cytoplasmic protein that participates in the assembly of cell-cell adherens junctions by binding cadherins to the actin cytoskeleton. In addition, it is a key component of the Wnt signaling pathway. Activation of this pathway triggers the accumulation of beta-catenin in the nucleus, where it activates the transcription of target genes. Abnormal accumulation of beta-catenin is characteristic of various types of cancer and is caused by mutations either in the adenomatous polyposis coli protein, which regulates beta-catenin degradation, or in the beta-catenin molecule itself. Aberrant accumulation of beta-catenin in tumors is often associated with mutational inactivation of the p53 tumor suppressor. Here we show that overexpression of wild-type p53, by either transfection or DNA damage, down-regulates beta-catenin in human and mouse cells. This effect was not obtained with transcriptionally inactive p53, including a common tumor-associated p53 mutant. The reduction in beta-catenin level was accompanied by inhibition of its transactivation potential. The inhibitory effect of p53 on beta-catenin is apparently mediated by the ubiquitin-proteasome system and requires an active glycogen synthase kinase 3beta (GSK3beta). Mutations in the N terminus of beta-catenin which compromise its degradation by the proteasomes, overexpression of dominant-negative DeltaF-beta-TrCP, or inhibition of GSKbeta activity all rendered beta-catenin resistant to down-regulation by p53. These findings support the notion that there will be a selective pressure for the loss of wild-type p53 expression in cancers that are driven by excessive accumulation of beta-catenin.
Figures




References
-
- Abarzua P, LoSardo J E, Gubler M L, Neri A. Microinjection of monoclonal antibody PAb421 into human SW480 colorectal carcinoma cells restores the transcription activation function to mutant p53. Cancer Res. 1995;55:3490–3494. - PubMed
-
- Adams C, Nelson W. Cytomechanics of cadherin-mediated cell-cell adhesion. Curr Opin Cell Biol. 1998;10:572–577. - PubMed
-
- Albrechtsen N, Dornreiter I, Grosse F, Kim E, Wiesmuller L, Deppert W. Maintenance of genomic integrity by p53: complementary roles for activated and non-activated p53. Oncogene. 1999;18:7706–7717. - PubMed
-
- Ashcroft M, Vousden K H. Regulation of p53 stability. Oncogene. 1999;18:7637–7643. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
