Age-dependent cognitive deficits and neuronal apoptosis in cyclooxygenase-2 transgenic mice
- PMID: 11588192
- PMCID: PMC6763862
- DOI: 10.1523/JNEUROSCI.21-20-08198.2001
Age-dependent cognitive deficits and neuronal apoptosis in cyclooxygenase-2 transgenic mice
Abstract
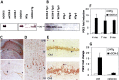
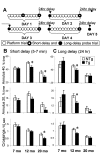
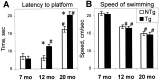
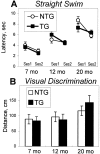
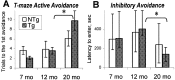
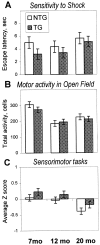
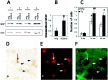
The cyclooxygenases catalyze the rate-limiting step in the formation of prostaglandins from arachidonic acid and are the pharmacological targets of (NSAIDs). In brain, cyclooxygenase-2 (COX-2), the inducible isoform of cyclooxygenase, is selectively expressed in neurons of the cerebral cortex, hippocampus, and amygdala. As an immediate-early gene, COX-2 is dramatically and transiently induced in these neurons in response to NMDA receptor activation. In models of acute excitotoxic neuronal injury, elevated and sustained levels of COX-2 have been shown to promote neuronal apoptosis, indicating that upregulated COX-2 activity is injurious to neurons. COX-2 may also contribute to the development of Alzheimer's disease, for which early administration of NSAIDs is protective against development of the disease. To test the effect of constitutively elevated neuronal COX-2, transgenic mice were generated that overexpressed COX-2 in neurons and produced elevated levels of prostaglandins in brain. In cross-sectional behavioral studies, COX-2 transgenic mice developed an age-dependent deficit in spatial memory at 12 and 20 months but not at 7 months and a deficit in aversive behavior at 20 months of age. These behavioral changes were associated with a parallel age-dependent increase in neuronal apoptosis occurring at 14 and 22 months but not at 8 months of age and astrocytic activation at 24 months of age. These findings suggest that neuronal COX-2 may contribute to the pathophysiology of age-related diseases such as Alzheimer's disease by promoting memory dysfunction, neuronal apoptosis, and astrocytic activation in an age-dependent manner.
Figures
References
-
- Aigner L, Arber S, Kapfhammer JP, Laux T, Schneider C, Botteri F, Brenner HR, Caroni P. Overexpression of the neural growth-associated protein GAP-43 induces nerve sprouting in the adult nervous system of transgenic mice. Cell. 1995;83:269–278. - PubMed
-
- Bailey CH, Montarolo PG, Chen M, Kandel ER, Schacher S. Inhibitors of protein and RNA synthesis block the structural changes that accompany long-term facilitation in Aplysia. Neuron. 1992;9:749–758. - PubMed
-
- Barnes CA. Aging and the physiology of spatial memory. Neurobiol Aging. 1988;9:563–568. - PubMed
-
- Bezzi P, Carmignoto G, Pasti L, Vesce S, Rossi D, Rizzini BL, Pozzan T, Volterra A. Prostaglandins stimulate calcium dependent glutamate release in astrocytes. Nature. 1998;391:281–285. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
